Abstract
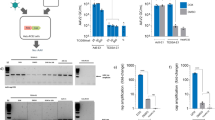
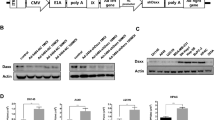
A critical issue for E1-deleted adenoviral vectors manufactured from 293 cells is the emergence of replication-competent adenovirus (RCA). These contaminants arise through homologous recombination between identical sequences framing the E1 locus displayed by 293 cells, and the vector backbones. Modified recombinogenic sequences (syngen) were thus introduced within the vector backbone, and virus viability and RCA emergence were assessed. Syngen#1 is a synthetic sequence displaying silent point mutations in the pIX and IVa2 coding regions. A side by side comparison of Ad5CMV/p53 (E1-deleted adenovirus expressing the p53 tumor suppressor gene) and AVΔE1#1CMV/p53 (with syngen#1 in place of wild-type sequences) demonstrated a normal productivity for the modified construct. The altered sequences did not impair p53-mediated apoptosis in a model tumor cell line. Most importantly, a statistically significant decrease in terms of RCA occurrence could also be demonstrated. Degenerescence of the recombinogenic sequences could be further accentuated by modifying non-coding pIX region (syngen #2), with no effect on virus productivity and stability. We concluded that these vector modifications constitute a feasible strategy to reduce RCA emergence during amplification in 293 cells. This approach could also be applied to decrease reincorporation of the E1 genes during amplification of ΔE1ΔE4 vectors in 293/E4-transcomplementing cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Stratford-Perricaudet LD, Perricaudet M . Gene transfer into animals: the promise of adenovirus Cohen-Adenauer O, Boiron M (eds); Human Gene Transfer John Libbey Eurotext 1991 51–61
Graham FL, Smiley J, Russel WC, Nairn R . Characteristics of a human cell line transformed by DNA from human adenovirus type 5 J Gen Virol 1977 36: 59–72
Louis N, Evelegh C, Graham FL . Cloning and sequencing of the cellular-viral junctions from the human adenovirus type 5 transformed 293 cell line Virology 1997 233: 423–429
Lochmüller H et al. Emergence of early region 1-containing replication-competent adenovirus in stocks of replication-defective adenovirus recombinant (ΔE1+ΔE3) during multiple passages in 293 cells Hum Gene Ther 1994 5: 1485–1491
Hehir KM et al. Molecular characterization of replication-competent variants of adenovirus vectors and genome modifications to prevent their occurence J Virol 1996 70: 8459–8467
Zhu J et al. Characterization of replication-competent adenovirus isolates from large-scale production of a recombinant adenoviral vector Hum Gene Ther 1999 10: 113–121
Hermens WT, Verhaagen J . Adenoviral vector-mediated gene expression in the nervous system of immunocompetent Wistar and T cell-deficient nude rats Hum Gene Ther 1997 8: 1049–1063
Murphy DB . Guidance for Human somatic cell therapy and gene therapy Hum Gene Ther 1998 9: 1513–1524
Fallaux FJ et al. New helper cells and matched early region 1-deleted adenovirus vectors prevent generation of repication-competent adenoviruses Hum Gene Ther 1998 9: 1909–1917
Dedieu JF et al. Long term gene delivery into livers of immunocompetent mice with E1/E4-defective adenoviruses J Virol 1997 71: 4626–4637
Holliday R . A mechanism for gene conversion in fungi Genet. Res 1964 5: 282–304
Medelson MS, Radding CM . A general model for genetic recombination. Proc Natl Acad Sci USA 1975 72: 358–361
Szostack JW, Orr-Weaver TL, Rothstein RJ, Stahl FW . The double-strand-break repair model for recombination Cell 1983 33: 25–35
Rubnitz J, Subramani S . The minimum amount of homology required for homologous recombination in mammalian cells Mol Cell Biol 1884 4: 2253–2258
Waldman AS, Liskay RM . Differential effects of base-pair mismatch on intrachromosomal versus extrachromosomal recombination in mouse cells Proc Natl Acad Sci USA 1987 84: 5340–5344
TeRiele H, Maandag ER, Berns A . Highly efficient gene targeting in embrionic stem cells through homologous recombination with isogenic DNA constructs Proc Natl Acad Sci USA 1992 89: 5128–5132
Crouzet J et al. Recombinational construction in Escherichia coli of infectious adenoviral genome Proc Natl Acad Sci USA 1997 94: 1414–1419
Lutz P et al. The product of the adenovirus intermediate gene IX is a transcriptional activator J Virol 1997 71: 5102–5109
Dijkema R et al. Gene organization of the transforming region of the adenovirus type 7 Gene 1982 18: 143–156
Liskay RM, Letsou A, Stachelek JL . Homology requirement for efficient gene conversion between duplicated chromosomal sequences in mammalian cells Genetics 1987 115: 161–167
Ayares D, Chekuri L, Song K-Y, Kucherlapati R . Sequence homology requirements for intermolecular recombination in mammalian cells Proc Natl Acad Sci USA 1986 83: 5199–5203
Yeh P et al. Efficient dual transcomplementation of adenovirus E1 and E4 regions from a 293-derived cell line expressing a minimal E4 functional unit J Virol 1996 70: 559–565
Marsh JL, Erfle M, Wykes EJ . The pIC plasmid and phage vectors with versatile cloning sites for recombinant selection by insertional inactivation Gene 1984 32: 481–485
Chroboczek J, Bieder F, Jacrot B . The sequence of the genome of the adenovirus type 5 and its comparison with the genome of the adenovirus type 2 Virology 1992 186: 280–285
Zhang WW et al. High efficiency gene transfer and high level expression of wild type p53 in human lung cancer cells mediated by recombinant adenovirus Cancer Gene Ther 1994 1: 5–13
Weinberg DH, Ketner G . A cell line that supports the growth of a defective early region 4 deletion mutant in human adenovirus type 2 Proc Natl Acad Sci USA 1983 80: 5383–5386
Robert JJ, Geoffroy MC, Finiels F, Mallet J . An adenoviral vector-based system to study neuronal gene expression: Analysis of the rat tyrosine hydroxylase promoter in cultured neurons J Neurochem 1997 68: 2152–2160
Cramér H . Mathematical Methods of Statistics Princeton University Press 1946
Acknowledgements
The authors thank Claude Kedinger for anti-pIX and anti-IVa2 antibodies, F Bellot for anti-virion L5 antibodies and T Huet for pIN116 plasmid.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Robert, JJ., Gauffeny, I., Maccario, J. et al. Degenerated pIX-IVa2 adenoviral vector sequences lowers reacquisition of the E1 genes during virus amplification in 293 cells. Gene Ther 8, 1713–1720 (2001). https://doi.org/10.1038/sj.gt.3301562
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301562