Abstract
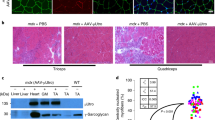
Skeletal muscle is a privileged target for long-term rAAV-mediated gene transfer in mouse, rat, dog and non-human primates. Intramuscular injections of rAAV encoding human factor IX in hemophilia B patients have been initiated, based on promising results gathered in affected dogs. We found that intramuscular rAAV administration in rats resulted in restricted transduction essentially along the myofibers axis with poor lateral diffusion. This suggested that the transduction rate might be limited by the ability of the virus to reach sites distant from the injection point. We tested whether hyaluronidase, an enzyme which dissociates the extracellular matrix, could enhance vector diffusion when injected in the rat muscle before administration of rAAV encoding either nuclear-localized β-galactosidase (rAAVCMVnlsLacZ) or the human α-1-antitrypsin (rAAVCMVhAAT) under the control of the cytomegalovirus immediate–early promoter (CMV). The results showed that pretreatment of the rat anterior tibialis muscle with hyaluronidase resulted in: (1) a larger diffusion of the virus indicated by an increase in the area containing LacZ-transduced fibers, and (2) a two- to three-fold increase of transduction efficiency measured by the number of LacZ-positive fibers or by the hAAT serum concentration. We also provide evidence that hyaluronidase was well tolerated and was not associated with short- or long-term toxicity evaluated by morphological studies. Finally, in our experimental conditions, hyaluronidase did not promote rAAV dissemination to other organs as assessed by PCR to detect vector sequences. We conclude that pretreament of skeletal muscle by hyaluronidase, a clinically available reagent, was harmless and resulted in a consistent and significant increase in rAAV diffusion and transduction levels.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Snyder RO . Adeno-associated virus-mediated gene delivery J Gene Med 1999 1: 166–175
Grimm D, Kleinschmidt JA . Progress in adeno-associated virus type 2 vector production: promises and prospects for clinical use Hum Gene Ther 1999 10: 2445–2450
Bennett J et al. Stable transgene expression in rod photoreceptors after recombinant adeno-associated virus-mediated gene transfer to monkey retina Proc Natl Acad Sci USA 1999 96: 9920–9925
Herzog RW et al. Long-term correction of canine hemophilia B by gene transfer of blood coagulation factor IX mediated by adeno-associated viral vector Nature Med 1999 5: 56–63
Haberman RP, McCown TJ, Samulski RJ . Inducible long-term gene expression in brain with adeno-associated virus gene transfer Gene Therapy 1998 5: 1604–11
Snyder RO et al. Efficient and stable adeno-associated virus-mediated transduction in the skeletal muscle of adult immunocompetent mice Hum Gene Ther 1997 8: 1891–1900
Xiao X, Li J, Samulski RJ . Efficient long-term gene transfer into muscle tissue of immunocompetent mice by adeno-associated virus vector J Virol 1996 70: 8098–8108
Song S et al. Sustained secretion of human alpha-1-antitrypsin from murine muscle transduced with adeno-associated virus vectors Proc Natl Acad Sci USA 1998 95: 14384–8
Salvetti A et al. Factors influencing recombinant adeno-associated virus production Hum Gene Ther 1998 9: 695–706
Fromes Y et al. Gene delivery to the myocardium by intrapericardial injection Gene Therapy 1999 6: 683–688
Acknowledgements
We are indebted to Estelle Toublanc and the Vector Core of the University Hospital of Nantes for providing the rAAVCMVnlsLacZ and the rAAVCMVhAAT vectors. We also thank Lydie Guigand for technical support for the morphological studies. This work was supported by grants from the Association Francaise contre les Myopathies (AFM) and the Association Nantaise de Thérapie Génique (ANTG).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Favre, D., Cherel, Y., Provost, N. et al. Hyaluronidase enhances recombinant adeno-associated virus (rAAV)-mediated gene transfer in the rat skeletal muscle. Gene Ther 7, 1417–1420 (2000). https://doi.org/10.1038/sj.gt.3301256
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301256
Keywords
This article is cited by
-
Induction of a local muscular dystrophy using electroporation in vivo: an easy tool for screening therapeutics
Scientific Reports (2020)
-
Gene Therapy for the Nervous System: Challenges and New Strategies
Neurotherapeutics (2014)
-
Gene Transfer: How Can the Biological Barriers Be Overcome?
The Journal of Membrane Biology (2010)
-
Non-viral gene delivery in skeletal muscle: a protein factory
Gene Therapy (2003)
-
Recombinant adeno-associated virus type 2 mediates highly efficient gene transfer in regenerating rat skeletal muscle
Gene Therapy (2002)