Abstract
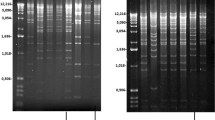
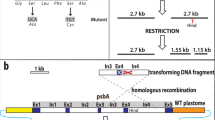
In the absence of a combined nitrogen source, such as ammonia, approximately every tenth vegetative cell along filaments of the cyanobacterium Anabaena develops into a heterocyst, a terminally differentiated cell that is morphologically and biochemically specialized for nitrogen fixation1. At least two specific DNA rearrangements involving the nitrogen-fixation (nif) genes occur during heterocyst differentiation2, one within the nifD gene and the other near the nifS gene. The two rearrangements have several properties in common. Both occur quantitatively in all heterocyst genomes, both occur at approximately the same developmental time, late in the process of heterocyst differentiation2, and both result from site-specific recombination between short repeated DNA sequences. We report here the nucleotide sequences found at the site of recombination near the nifS gene. These sequences differ from those found previously for the nifD rearrangement2, suggesting that the two rearrangements are catalysed by different enzymes and may be regulated independently. We also show that the nifS gene is transcribed only from rearranged genomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
1. Haselkorn, R. A. Rev. PL Physiol. 29, 319–344 (1978). 2. Golden, J. W., Robinson, S. J. & Haselkorn, R. Nature 314, 419–423 (1985). 3. Haselkorn, R., Golden, J. W., Lammers, P. J. & Mulligan, M. E. in Nitrogen Fixation Research Progress (eds Evans, H. J., Bottomly, P. J. & Newton, W. E.) 485–490 (Nijhoff, Dordrecht, 1985). 4. Lammers, P. J., Golden, J. W. & Haselkorn, R. Cell 44, 905–911 (1986). 5. Rice, D., Mazur, B. J. & Haselkorn, R. J. biol. Chem. 257, 13157–13163 (1982). 6. Davis, R. W., Botstein, D. & Roth, J. R. Advanced Bacterial Genetics (Cold Spring Harbor Laboratory, New York, 1980). 7. Soberon, X., Covarrubias, L. & Bolivar, F. Gene 9, 287–305 (1980). 8. Loenen, W. A. M. & Brammar, W. J. Gene 10, 249–259 (1980). 9. Yanisch–Perron, C, Vieira, J. & Messing, J. Gene 33, 103–119 (1985). 10. Buikema, W. J., Klingensmith, J. A., Gibbons, S. L. & Ausubel, F. M. J. Bact. 169,1120–1126 (1987). 11. Noti, J. D., Folkerts, O., Turken, A. N. & Szalay, A. A. /. Bact. 167, 774–783 (1986). 12. Maniatis, T., Fritsch, E. F. & Sambrook, J. Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Laboratory, New York, 1982). 13. Vogelstein, B. & Gillespie, D. Proc. natn. Acad. Sci. U.S.A. 76, 615–619 (1979). 14. Maxam, A. M. & Gilbert, W. Meth. Enzym. 65, 499–560 (1980). 15. Turner, N. E., Robinson, S. J. & Haselkorn, R. Nature 306, 337–342 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Golden, J., Mulligan, M. & Haselkorn, R. Different recombination site specificity of two developmentally regulated genome rearrangements. Nature 327, 526–529 (1987). https://doi.org/10.1038/327526a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/327526a0
This article is cited by
-
Excision of the nifD element in the heterocystous cyanobacteria
Archives of Microbiology (2008)
-
Characterization of a 4 kb Variant of the nifD Element inAnabaena sp. Strain ATCC 33047
Current Microbiology (2005)
-
A model for cell type-specific differential gene expression during heterocyst development and the constitution of aerobic nitrogen fixation ability inAnabaena sp. strain PCC 7120
Journal of Biosciences (1996)
-
Rearrangements of nitrogen fixation (nif) genes in the heterocystous cyanobacteria
J Biosci (1994)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.