Abstract
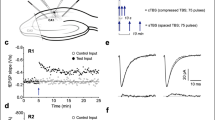
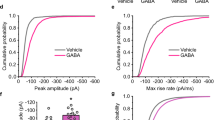
Activity-dependent alteration in synaptic strength is a fundamental property of the vertebrate central nervous system and is thought to underlie learning and memory. The most extensively studied model of activity-dependent synaptic plasticity is long-term potentiation (LTP) of glutamate-responsive (glutamatergic) synapses, a widespread phenomenon involving multiple mechanisms1. The best characterized form of LTP occurs in the CA1 region of the hippocampus, in which LTP is initiated by transient activation of NMDA (N-methyl-D-aspartate) receptors and is expressed as a persistent increase in synaptic transmission through AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionate) receptors2. This increase is due, at least in part, to a postsynaptic modification of AMPA-receptor function3; this modification could be caused by an increase in the number of receptors, their open probability, their kinetics or their single-channel conductance. Here we show that the induction of LTP in the CA1 region of the hippocampus is often associated with an increase in single-channel conductance of AMPA receptors. This shows that elementary channel properties can be rapidly modified by synaptic activity and provides an insight into one molecular mechanism by which glutamatergic synapses can alter their strength.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bliss, T. V. P. & Collingridgee, G. L. Asynaptic model of memory: long-term potentiation in the hippocampus. Nature 361, 31–39 (1993).
Collingridge, G. L., Kehl, S. J. & McLennan, H. Excitatory amino acids in synaptic transmission in the Schaffer collateral-commissural pathway of the rat hippocampus. J. Physiol. (Lond.) 334, 33–46 (1983).
Davies, S. N., Lester, R. A. J., Reymann, K. G. & Collingridgee, G. L. Temporally distinct pre- and post-synaptic mechanisms maintain long-term potentiation. Nature 338, 500–503 (1989).
Traynelis, S. F., Silver, R. A. & Cull-Candy, S. G. Estimated conductance of glutamate receptor channels activated during EPSCs at the cerebellar mossy fiber-granule cell synapse. Neuron 11, 279–289 (1993).
De Koninck, Y. & Mody, I. Noise analysis of miniature IPSCs in adult rat brain slices: Properties and modulation of synaptic GABAAreceptor channels. Neurophysiol. 71, 1318–1335 (1994).
Silver, R. A., Cull-Candy, S. G. & Takahashi, T. Non-NMDA glutamate receptor occupancy and open probability at a rat cerebellar synapse with single and multiple release sites. J. Physiol. (Lond.) 494, 231–250 (1996).
Jonas, P., Major, G. & Sakmann, B. Quantal components of unitary EPSCs at the mossy fibre synapse on CA3 pyramidal cells of rat hippocampus. J. Physiol. (Lond.) 472, 615–663 (1993).
Spruston, N., Jonas, P. & Sakmann, B. Dendritic glutamate receptor channels in rat hippocampal CA3 and CA1 pyramidal neurons. J. Physiol. (Lond.) 482, 325–352 (1995).
Major, G., Larkman, A. U., Jonas, P., Sakmann, B. & Jack, J. J. B. Detailed passive cable models of whole-cell recorded CA3 pyramidal neurons in rat hippocampal slices. J. Neurosci. 14, 4613–4638 (1994).
Clay, J. R. & DeFelice, L. J. Relationship between membrane excitability and single channel open-close kinetics. Biophys. J. 42, 151–157 (1983).
Clements, J. D., Lester, R. A. J., Tong, G., Jahr, C. E. & Westbrook, G. L. The time course of glutamate in the synaptic cleft. Science 258, 1498–1501 (1992).
Stuart, G. J., Dodt, H.-U. & Sakmann, B. Patch-clamp recordings from the soma and dendrites of neurons in brain slices using infrared video microscopy. Pflugers Arch. 423, 511–518 (1993).
Malinow, R. & Tsien, R. W. Presynaptic enhancement shown by whole-cell recordings of long-term potentiation in hippocampal slices. Nature 346, 177–180 (1990).
Stevens, C. F. & Wang, Y. Changes in reliability of synaptic function as a mechanism for plasticity. Nature 371, 704–707 (1994).
Isaac, J. T. R., Hjelmstad, G. O., Nicoll, R. A. & Malenka, R. C. Long-term potentiation at single fiber inputs to hippocampal CA1 pyramidal cells. Proc. Natl Acad. Sci. USA 93, 8710–8715 (1996).
Allen, C. & Stevens, C. F. An evaluation of causes for unreliability of synaptic transmission. Proc. Natl Acad. Sci. USA 91, 10380–10383 (1994).
Swanson, G. T., Kamboj, S. K. & Cull-Candy, S. G. Single-channel properties of recombinant AMPA receptors depend on RNA editing, splice variation, and subunit composition. J. Neurosci. 17, 58–69 (1997).
Greengard, P., Jen, J., Nairn, A. C. & Stevens, C. F. Enhancement of the glutamate resposne by cAMP-dependent protein kinase in hippocampal neurones. Science 253, 1135–1138 (1991).
Wang, L.-Y., Salter, M. W. & MacDonald, J. F. Regulation of kainate receptors by cAMP-dependent protein kinase and phosphatases. Science 253, 1132–1135 (1991).
Cull-Candy, S. G. & Usowicz, M. M. Multiple-conductance channels activated by excitatory amino acids in cerebellar neurons. Nature 325, 525–528 (1987).
Jahr, C. E. & Stevens, C. F. Glutamate activates multiple single channel conductances in hippocampal neurons. Nature 325, 522–525 (1987).
Barria, A., Muller, D., Derkach, V., Griffith, L. C. & Soderling, T. R. Regulatory phosphorylation of AMPA-type glutamate receptors by CaM-KII during long-term potentiation. Science 276, 2042–2045 (1997).
Kullmann, D. M. & Siegelbaum, S. A. The site of expression of NMDA receptor-dependent LTP: new fuel for old fire. Neuron 15, 997–1002 (1995).
Shrike, A. M. & Malinow, R. Mechanisms of potentiation by calcium-calmodulin kinase II of postsynaptic sensitivity in rat hippocampal neurons. J. Neurophysiol. 78, 2682–2692 (1997).
Liao, D., Hessler, N. A. & Malinow, R. Activation of postsynaptically silent synapses during pairing-induced LTP in CA1 region of hippocampal slice. Nature 375, 400–404 (1995).
Isaac, J. T. R., Nicoll, R. A. & Malenka, R. C. Evidence for silent synapses: implications for the expression of LTP. Neuron 15, 427–434 (1995).
Traynelis, S. F. & Wahl, P. Control of rat GluR6 glutamate receptor open probability by protein kinase A and calcineurin. J. Physiol. (Lond.) 503, 513–531 (1997).
Hines, M. L. Efficient computation of branched nerve equations. Int. J. Biomed. Comp. 15, 69–76 (1984).
Homes, W. R. & Levy, W. B. Insights into associative long-term potentiation from computational models of NMDA receptor-mediated calcium influx and intracellular calcium concentration changes. J. Neurophysiol. 63, 1148–1168 (1990).
Sigworth, F. J. The variance of sodium current fluctuations at the node of Ranvier. J. Physiol. (Lond.) 307, 97–129 (1980).
Acknowledgements
We thank A. Silver and M. Farrant for their advice, and W. W. Anderson for providing the data acquisition software. This work was supported by the MRC (T.A.B., G.L.C.), NIH (T.A.B.), Swiss National Science Foundation (A.L.) and Wellcome Trust (J.T.R.I., G.L.C.).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Benke, T., Lüthi, A., Isaac, J. et al. Modulation of AMPA receptor unitary conductance by synaptic activity. Nature 393, 793–797 (1998). https://doi.org/10.1038/31709
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/31709
This article is cited by
-
Targeting metaplasticity mechanisms to promote sustained antidepressant actions
Molecular Psychiatry (2024)
-
CaMKII: a central molecular organizer of synaptic plasticity, learning and memory
Nature Reviews Neuroscience (2022)
-
On the Role of Speed in Technological and Biological Information Transfer for Computations
Acta Biotheoretica (2022)
-
PKA drives an increase in AMPA receptor unitary conductance during LTP in the hippocampus
Nature Communications (2021)
-
The glutamatergic synapse: a complex machinery for information processing
Cognitive Neurodynamics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.