Abstract
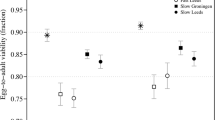
The importance of inbreeding depression in theoretical considerations of mating-system evolution1,2,3,4,5 and its potential impact on the persistence of small populations6 has renewed interest in the genetic basis of this phenomenon. Inbreeding increases homozygosity. This can produce inbreeding depression for two different reasons: first, deleterious recessive or partially recessive alleles that are masked at heterozygous loci by dominant alleles become fully expressed in homozygotes; and second, alleles may interact in an overdominant manner, such that the fitness of either type of homozygote is lower than that of heterozygotes. These two mechanisms produce different long-term effects in populations experiencing increased levels of inbreeding. Inbreeding depression resulting from deleterious alleles can be removed by selection, but inbreeding depression produced by overdominance cannot be removed without lowering the mean fitness of the population1,2,3,4,5. Using a North Carolina 3 breeding programme7, the most powerful quantitative genetics technique available8,9,10, we show here that deleterious recessive alleles are mainly responsible for inbreeding depression in two closely related annual plants, the primarily selfing Mimulus micranthus and the mixed-mating M. guttatus. Estimates indicate that deleterious alleles in M. micranthus are more nearly additive than they are in M. guttatus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Charlesworth, D. & Charlesworth, B. Inbreeding depression and its evolutionary consequences. Annu. Rev. Ecol. Syst. 18, 237–268 (1987).
Charlesworth, D., Morgan, M. T. & Charlesworth, B. Inbreeding depression, genetic load, and the evolution of outcrossing rates in a multilocus system with no linkage. Evolution 44, 1469–1489 (1990).
Lande, R. & Schemske, D. W. The evolution of self-fertilization and inbreeding depression in plants. I. Genetic models. Evolution 39, 24–40 (1985).
Holsinger, K. E. Inbreeding depression and the evolution of plant mating systems. Trends Ecol. Evol. 6, 307–308 (1991).
Uyenoyama, M. K., Holsinger, K. E. & Waller, D. M. in Oxford Surveys in Evolutionary Biology Vol. 9(eds Futuyma, D. & Antonovics, J.) 327–381 (Oxford Univ. Press, New York, (1993)).
Fenster, C. B. & Dudash, M. R. in Restoration of Endangered Species (eds Bowles, M. L. & Whelan, C. J.) 34–62 (Cambridge Univ. Press, Cambridge, UK, (1994)).
Comstock, R. E. & Robinson, H. F. in Heterosis (ed. Gowen, J. W.) 494–516 (Iowa State College Press, Ames, (1952)).
Jinks, J. L. in Heterosis: Reappraisal of Theory and Practice (ed. Frankel, R.) 1–46 (Springer, Berlin, (1983)).
Kearsey, M. J. The efficiency of the North Carolina Experiment III and the selfing backcrossing series for estimating additive and dominance variation. Heredity 45, 73–82 (1980).
Bulmer, M. G. The Mathematical Theory of Quantitative Genetics (Clarendon, Oxford, (1985)).
El-Nahrawy, M. A. & Bingham, E. T. Performance of S1 alfalfa lines from original and improved populations. Crop Sci. 29, 920–923 (1989).
Moll, R. H., Lindsey, M. F. & Robinson, H. F. Estimates of genetic variances and level of dominance in maize. Genetics 49, 411–423 (1964).
Hallauer, A. R. & Miranda, J. B. Quantitative Genetics in Maize Breeding (Iowa State Univ. Press, Ames, (1985)).
Apirion, D. & Zohary, D. Chlorophyll lethal in natural populations of the orchard grass (Dactylis glomerata L.). A case of balanced polymorphism in plants. Genetics 46, 393–399 (1961).
Williams, W. & Brown, A. G. Genetic response to selection in cultivated plants: gene frequencies in Prunus avium. Heredity 10, 237–245 (1956).
Gustafsson, A. The coorperation of genotypes in barley. Hereditas 39, 1–18 (1950).
Barrett, S. C. H. & Charlesworth, D. Effects of a change in the level of inbreeding on the genetic load. Nature 352, 522–524 (1991).
Johnston, M. O. & Schoen, D. J. Mutation rates and dominance levels of genes affecting total fitness in two angiosperm species. Science 267, 226–229 (1995).
Willis, J. H. Effects of different levels of inbreeding on fitness components in Mimulus guttatus. Evolution 47, 864–876 (1993).
Fenster, C. B. & Ritland, K. Chloroplast DNA and isozyme diversity in two Mimulus species (Scrophulariaceae) with contrasting mating systems. Am. J. Bot. 79, 1440–1447 (1992).
Fu, Y.-B. & Ritland, K. Evidence for the partial dominance of viability genes contributing to inbreeding depression in Mimulus guttatus. Genetics 136, 323–331 (1994).
Latta, R. & Ritland, K. The relationship between inbreeding depression and prior inbreeding among populations of four Mimulus taxa. Evolution 48, 806–817 (1994).
Carr, D. E. & Fenster, C. B. Levels of genetic variation and covaration for Mimulus (Scrophulariaceae) floral traits. Heredity 72, 606–618 (1994).
Carr, D. E. & Dudash, M. R. Inbreeding depression in two species of Mimulus (Scrophulariaceae) with contrasting mating systems. Am. J. Bot. 83, 586–593 (1996).
Dudash, M. R. & Ritland, K. Multiple paternity and self-fertilization in relation to floral age in Mimulus guttatus (Scrophulariaceae). Am. J. Bot. 78, 1746–1753 (1991).
Comstock, R. E. & Robinson, H. F. The components of genetic variation in populations of biparental progenies and their use in estimating the average degree of dominance. Biometrics 2, 254–266 (1948).
Husband, B. C. & Schemske, D. W. Evolution of the magnitude and timing of inbreeding depression in plants. Evolution 50, 54–70 (1996).
Dudash, M. R., Carr, D. E. & Fenster, C. B. Five generations of enforced selfing and outcrossing in Mimulus guttatus: inbreeding depression variation at the population and family level. Evolution 51, 54–65 (1997).
Melchinger, A. E., Geiger, H. H. & Schnell, F. W. Epistasis in maize (Zea mays L.) 2. Genetic effects in crosses among early flint and dent inbred lines determined by three methods. Theor. Appl. Genet. 72, 231–239 (1986).
Pray, L. A. & Goodnight, C. J. Genetic variation in inbreeding depression in the red flour beetle Tribolium castaneum. Evolution 49, 176–188 (1995).
Acknowledgements
We thank M. Lawrence and J. Crow for encouragement, J. Coors for statistical advice, and M. Bowers, L. Chao, D. Charlesworth, C. Cole, L. Delph, J. Dooley, C. Fenster and D.McCauley for comments on earlier versions of the manuscript. This research was supported by the NSF and Maryland Agricultural Experiment Station.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dudash, M., Carr, D. Genetics underlying inbreeding depression in Mimulus with contrasting mating systems. Nature 393, 682–684 (1998). https://doi.org/10.1038/31468
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/31468
This article is cited by
-
Fitness effects of somatic mutations accumulating during vegetative growth
Evolutionary Ecology (2022)
-
Mathematical modelling for variations of inbreeding populations fitness with single and polygenic traits
BMC Genomics (2017)
-
Self-fertilization and inbreeding depression in three ascidian species that differ in genetic dispersal potential
Marine Biology (2017)
-
Analysis of the effects of inbreeding on lifespan and starvation resistance in Drosophila melanogaster
Genetica (2011)
-
Evidence for inbreeding depression and post-pollination selection against inbreeding in the dioecious plant Silene latifolia
Heredity (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.