Abstract
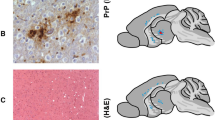
Scrapie is a slow infection of the nervous system which progresses in the absence of any apparent immune response1–8. The recent development of a large-scale purification protocol for scrapie prion9 made it possible to obtain substantial quantities of electrophoretically purified prion protein (PrP 27–30) and we report here on the successful production of a rabbit antiserum to PrP 27–30. The antiserum reacted with PrP 27–30 and several lower molecular weight proteins as shown by Western blots; it did not react with protein preparations from uninfected brains. Discrete structures in the subependymal region of scrapie-infected hamster brains were stained immunocytochemically. These same structures also stained with Congo red dye and showed green birefringence with polarized light, a characteristic of purified prion rods9. This staining pattern suggests that they are amyloid plaques.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Zlotnik, I. & Rennie, J. C. J. comp. Path. 75, 147–157 (1965).
Beck, E. & Daniel, P. M. in Slow Transmissible Diseases of the Nervous System Vol. 1 (eds Prusiner, S. B. & Hadlow, W. J.) 253–270 (Academic, New York, 1979).
McFarlin, D. E., Raff, M. C., Simpson, E. & Nehlsen, S. Nature 233, 336 (1971).
Gibbs, C. J. Jr Curr. Top. Microbiol. Immun. 40, 44–58 (1967).
Kingsbury, D. T., Smeltzer, D. A., Gibbs, C. J. Jr & Gajdusek, D. C. Infect. Immun. 32, 1176–1180 (1981).
Stites, D. P., Garfin, D. E. & Prusiner, S. B. in Slow Transmissible Diseases of the Nervous System Vol. 2 (eds Prusiner, S. B. & Hadlow, W. J.) 211–224 (Academic, New York, 1979).
Kasper, K. C., Bowman, K., Stites, D. P. & Prusiner, S. B. in Hamster Immune Responses in Infectious and Oncologic Diseases (eds Streilein, J. W., Hart, D. A., Stein-Streilein, J., Duncan, W. R. & Billingham, R. E.) 401–413 (Plenum, New York, 1981).
Kasper, K. C., Stites, D. P., Bowman, K. A., Panitch, H. & Prusiner, S. B. J. Neuroimmun. 3, 187–201 (1982).
Prusiner, S. B. et al. Biochemistry 21, 6942–6950 (1982).
Bolton, D. C., McKinley, M. P. & Prusiner, S. B. Science 218, 1309–1311 (1982).
McKinley, M. P., Bolton, D. C. & Prusiner, S. B. Cell 35, 57–62 (1983).
Prusiner, S. B. et al. Cell 35, 349–358 (1983).
Diringer, H. et al.. Nature 306, 476–478 (1983).
Hawkes, R., Niday, E. & Gordon, J. Analyt. Biochem. 119, 142–147 (1982).
Burnette, W. N. Analyt. Biochem. 112, 195–203 (1981).
Towbin, H., Straehelin, T. & Gordon, J. Proc. natn. Acad. Sci. U.S.A. 76, 4350–4354 (1979).
Sternberger, L. A. in Immunocytochemistry 2nd edn, 104–169 (Wiley, New York, 1979).
Beck, E. & Daniel, P. M. in Slow, Latent and Temperate Virus Infections, NINDB Monogr. 2 (eds Gajdusek, D. C., Gibbs, C. J. Jr & Alpers, M.) 203–206 (U.S. Government Printing Office, Washington DC, 1965).
Bruce, M. E., Dickinson, A. G. & Fraser, H. Neuropath. appl. Neurobiol. 2, 471–478 (1976).
Baringer, J. R., Bowman, K. A. & Prusiner, S. B. J. Neuropath. exp. Neurol. 40, 329 (1981).
Tateishi, J., Nagara, H., Hikita, K. & Sato, Y. Ann. Neurol. 15, 278–280 (1984).
Mountjoy, C. Q., Tomlinson, B. E. & Gibson, P. H. J. neurol. Sci. 57, 89–103 (1982).
Gajdusek, D. C. Science 197, 943–960 (1977).
Hadlow, W. J., Prusiner, S. B., Kennedy, R. C. & Race, R. E. Ann. Neurol. 8, 628–631 (1980).
Prusiner, S. B. & Kingsbury, D. T. CRC Crit. Rev. clin. Neurobiol. (in the press).
Prusiner, S. B. Science 216, 136–144 (1982).
Laemmli, U. K. Nature 227, 680–685 (1970).
Sigel, M. B., Shina, Y. N. & Vanderlaan, W. P. in Meth. Enzym. 93, 3–12 (1983).
Bolton, A. E. & Hunter, W. M. Biochem. J. 133, 529–539 (1973).
Marsh, R. & Kimberlin, R. J. infect. Dis. 131, 104–110 (1975).
Puchtler, H., Sweaf, F. & Levin, M. J. Histochem. Cytochem. 10, 355–364 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bendheim, P., Barry, R., DeArmond, S. et al. Antibodies to a scrapie prion protein. Nature 310, 418–421 (1984). https://doi.org/10.1038/310418a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/310418a0
This article is cited by
-
New insights into early sequential PrPsc accumulation in scrapie infected mouse brain evidenced by the use of streptomycin sulfate
Histochemistry and Cell Biology (2008)
-
Amyloidosis in transgenic mice expressing murine amyloidogenic apolipoprotein A-II (Apoa2)
Laboratory Investigation (2007)
-
Differences in Proteinase K Resistance and Neuronal Deposition of Abnormal Prion Proteins Characterize Bovine Spongiform Encephalopathy (BSE) and Scrapie Strains
Molecular Medicine (1999)
-
Protein-glycosaminoglycan interactions: infectiological aspects
Medical Microbiology and Immunology (1996)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.