Abstract
Objective:
To report a case of Carbamazepine toxicity following the administration of Oxybutynin and Dantrolene.
Study design:
A case report.
Setting:
The Spinal Rehabilitation Department, Loewenstein Hospital, Raanana, Israel.
Methods:
A patient with C6D tetraplegia who sustained intoxication because of drug interaction is presented. She had been treated by Carbamazepine 1000 mg/day for neuropathic pain for 2 years without clinical or laboratory signs of toxicity. After administration of Oxybutynin concomitantly with an increase in the dose of Dantrolene, she presented the clinical symptoms and laboratory finding of Carbamazepine intoxication. Trying to adjust the treatment to the patient's requirements, Carbamazepine together with Oxybutynin and Dantrolene was readministrated in lower doses.
Results:
The combination of these drugs, even small doses, caused toxicity. Adding Dantrolene and Oxybutynin elevated the blood level of Carbamazepine, possibly by inhibition of cytochrome P450.
Conclusion:
A possible pharmacokinetic interaction between Dantrolene and Oxybutynin should be borne in mind when considering Carbamazepine medication for a patient with a spinal cord lesion.
Similar content being viewed by others
Introduction
Both Oxybutynin chloride and Dantrolene sodium are widely used in patients with spinal cord lesion (SCL). Oxybutynin is used for the treatment of detrusor overactivity1 and Dantrolene to reduce spasticity.2, 3, 4 Oxybutynin has both an antimuscarinic and a direct muscle relaxant effect and, in addition, a local anesthetic activity.5 It may have anticholinergic side effects, including dry mouth, blurred vision, somnolence, constipation, and delirium.6 Dantrolene relaxes striated muscles by inhibiting the release of Ca2+ from the sarcoplasmic reticulum.7 The drug commonly causes transient drowsiness, weakness, general malaise, fatigue, diarrhea, and liver test dysfunction that requires strict laboratory follow-up.8 The potential liver toxicity discourages some physicians from using Dantrolene, but still many use it for long periods.3, 4 Carbamazepine is less frequently used in patients with SCL, but it may be required when patients have concomitant post-traumatic or postoperative epilepsy, or more frequently for the treatment of painful or paraesthetic phenomena.9, 10 Carbamazepine intoxication includes nausea, vomiting, dizziness, ataxia, confusion, dysarthria, and nystagmus,11 and it may also cause liver dysfunction.12
Administration of the three drugs at the same time exposes the patient with SCL to their combined side effects. However, the reported case may indicate that the combination may carry an even greater risk because of pharmacokinetic interactions. Carbamazepine may interact with various drugs. It is eliminated through the hepatic pathway and catalyzed by cytochrome P450 (CYP3A subfamily),13 so drugs that inhibit the activity of this cytochrome may elevate Carbamazepine blood level and cause toxicity.11 These drugs include itriconazole, ketokonazole, clarythromycin, erythromycin, nefazodone, ritonavir,14 isoniazide, propoxyphene, cimetidine,15 fluvoxamine, fluoxetine,16 and acetazolamide.17 Here we present a case of Carbamazepine toxicity related to a pharmacokinetic interaction with either Oxybutynin or Dantrolene. To the best of our knowledge, this is the first such report.
Case report
A 37-year-old woman with incomplete tetraplegia due to cervical spondylosis at the C5–C6 level was admitted to the spinal department of Loewenstein Rehabilitation Hospital (Raanana, Israel). At 2 years before this admission she had had a subtotal excision of a D5–D7 ependymoma. Since that operation, she suffered from lancinating and burning sensations over the D6–D7 cutaneous dermatomes. Treatment with Gabapentin was unsuccessful, but the patient responded favorably to Tab. Carbamazepine 1000 mg daily. She was also receiving Tab. Baclofen 100 mg/day for spasticity, Tab. Atenolol 50 mg/day for hypertension, and Tab. Fluvoxamine 100 mg/day for depression. Neurological examination revealed spastic tetraplegia (ASIA D) below the C6 level, with exaggerated tendon reflexes and bilateral pyramidal signs. She needed assistance in self-care, could walk with crutches for a short distance, was voiding by self-catheterization, and had regular bowel movements. Between catheterizations, urine was leaking into the diaper. Admission laboratory examinations, including renal (BUN and creatinine) and liver (SGOT, SGPT, GGT) function tests, were normal. Abdominal sonogram revealed a normal liver and no other abnormalities.
Owing to the marked spasticity, Cap. Dantrolene was started and its dosage was elevated gradually. After urodynamic investigation revealed a hypertonic bladder without vesicoureteral reflux, Tab. Oxybutynin 5 mg twice a day was added.
At 2 weeks starting Oxybutynin, and while on 125 mg Dantrolene, the patient complained of dizziness and repeated vomiting. She was drowsy, unsteady, and confused, and her speech was slurred. Bilateral horizontal gaze-evoked nystagmus was found. The EEG record showed a slow background activity compatible with a toxic-metabolic pattern. Hemoglobin, leucocytes, blood electrolytes, liver, and renal function were normal, and Carbamazepine blood concentration was 16 μg/ml (therapeutic range 4–12 μg/ml).
Carbamazepine, Oxybutynin, Fluvoxamine, Baclofen, and Dantrolene were stopped, the plasma level of Carbamazepine returned to the therapeutic range (8.3 μg/ml), and the clinical condition of the patient gradually improved until she regained her previous status. Fluvoxamine, which may elevate the Carbamazepine plasma level, was discontinued permanently. However, as the pain, urinary frequency, and disturbing spasticity reappeared, Carbamazepine, Oxybutynin, and Dantrolene were readministered in doses of 600, 10, and 100 mg daily, respectively. As a Carbamazepine blood level of 9.2 μg/ml was reported under this treatment, and spasticity was still disturbing, Dantrolene dosage was raised to 125 mg daily. After 1 day, the Carbamazepine blood level increased to 29 μg/ml and clinical intoxication was again evident. Again, blood count, blood electrolytes, renal function test, and liver function test were normal. Carbamazepine and Oxybutynin were discontinued and Dantrolene dose was lowered to 25 mg per day. Carbamazepine level fell to 6 μg/ml and the intoxication disappeared, but then the patient was willing to use these drugs again to relieve her symptoms. Therefore, readministration of the medication was attempted again, using a lower dose: Carbamazepine 400 mg per day and Dantrolene 25 mg/day. After a week on this regimen the Carbamazepine blood level was 8.4 μg/ml. At this stage, 5 mg of Oxybutynin were added; it was followed by a dramatic elevation of Carbamazepine level to 32 μg/ml, and signs of toxicity returned. Liver enzymes remained within normal limits. This time Carbamazepine was replaced by Valproic acid 600 mg/day, which also proved to be beneficial against the painful sensation and showed no interactions with Oxybutynin or Dantrolene. The patient was discharged from hospital in an alert state and well oriented, independent in self-care, and walking with crutches.
Discussion
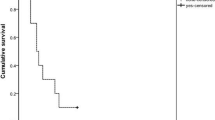
The clinical presentation and the laboratory finding of the patient presented here are compatible with Carbamazepine toxicity (Figure 1). As no signs of Carbamazepine overdose were evident during the previous 2 years, during which she received the same dosage of the drug, it is plausible that administration of new drugs changed the metabolism of Carbamazepine and elevated its blood level concentration.
The drug combinations: (1) Carbamazepine 1000 mg/day, Oxybutinine 10 mg/day, Dantrolene 125 mg/day; (2) none; (3) Carbamazepine 600 mg/day, Oxybutinine 10 mg/day, Dantrolene 100 mg/day; (4) Carbamazepine 600 mg/day, Oxybutinine 10 mg/day, Dantrolene 125 mg/day; (5) Carbamazepine 400 mg/day, Dantrolene 25 mg/day; (6) Carbamazepine 400 mg/day, Oxybutinine 5 mg/day, Dantrolene 25 mg/day. The first episode of intoxication followed the start of Oxybutynin and an increase in Dantrolene dosage to 125 mg/day. The second episode followed Oxybutinine administration with a similar increase in Dantrolene dosage. The third episode appeared with a lower dose of Dantrolene, when a low dose of Oxybutynin was started
During the previous 2 years she was taking, besides Carbamazepine, Baclofen, Atenolol, and Fluvoxamine. There is no evidence that Baclofen or Atenolol can increase Carbamazepine blood concentration. Fluvoxamine can do it,16 but it did not cause Carbamazepine toxicity in this patient during the previous 2 years, and signs of Carbamazepine overdose were evident after Fluvoxamine was stopped. Therefore, Fluvoxamine is unlikely to have participated in the intoxication.
Oxybutynin and Dantrolene were the only newly added drugs. Although Dantrolene and Carbamazepine have potential liver toxicity, liver function tests remained normal throughout the described period, and interaction with Oxybutynin, Dantrolene, or both is probably responsible for the intoxication. The appearance of intoxication after repeated challenges strongly supports this notion. It seems more plausible that Oxybutinine is responsible for the toxicity: Carbamazepine toxicity has never been demonstrated without concomitant Oxybutinine administration, and the first and third episodes of intoxication appeared when Oxybutynin was started. However, all the challenges included Dantrolene administration, and the second episode of intoxication occurred after Dantrolene dose was increased (Figure 1). Therefore, the contribution of Dantrolene to the toxicity cannot be excluded.
Existing reports about the metabolism of the drugs support the possibility of interaction with Carbamazepine. Several studies demonstrated that Oxybutynin is metabolized by oxidation via cytochrome P450 (CYP3A subfamily),18, 19 which also catalyzes Carbamazepine and influences activities of this and other cytochromes in human liver microsomes.20 The exact pathway of Dantrolene metabolism is unknown, but some reports suggest that Dantrolene decreases the activity of cytochrome P45021 and that this inhibition is dose dependent.22 The appearance of Carbamazepine toxicity with higher doses of Dantrolene after the first challenge implies that this interaction may also be dose dependent and may be due to cytochrome P450 inhibition. This implication is supported by the fact that Valproic acid, which is also used for treatment of neuropathic pain23 and is exclusively metabolized by microsomal glucoronide conjugation,24 did not interact with the P450-dependent drugs. However, it is not supported by the higher Carbamazepine blood level after the last challenge with the lower doses, and this ‘aquired sensitivity’ to the drug combination still requires an explanation. Therefore, further pharmacokinetic studies are needed to clarify the nature of the described interaction.
Conclusion
As Carbamazepine and Dantrolene are widely used in patients with SCL, physicians should pay attention to their possible interactions with drugs such as Carbamazepine. When an additional medication is necessary, alternative agents with lower interaction potential should be considered and dosage should be adjusted carefully, using serum drug concentration monitoring and a meticulous clinical observation to reduce the risk associated with interactions.
References
Gupta S, Sathyan G, Mori T . New perspectives on the overactive bladder: pharmacokinetics and bioavailability. Urology 2002; 60: 78–80.
Davidoff RA . Antispasticity drugs: mechanisms of action. Ann Neurol 1985; 17: 16–17.
Rode G et al. Medical treatment of spasticity. Neurochirurgie 2003; 49: 247–255.
Tarricco M, Adone R, Pagliacci C, Telaro E . Pharmacological intervention for spasticity following spinal cord injury. Cochrane Database Syst Rew 2000; 2: CD001131.
Andersson KE, Chapple CR . Oxybutynin and overactive bladder. World J Urol 2001; 19: 319–323.
Garely AD, Burrows LJ . Current pharmacotheraputic strategies for overactive bladder. Expert Opin Pharmacother 2002; 3: 827–833.
Fruen BR, Mickelson JR, Louis CF . Dantrolene inhibition of sarcoplasmic reticulum Ca2+ release by direct and specific action at skeletal muscle ryanodine receptors. J Biol Chem 1997; 272: 26965–26971.
Pinder RM, Brogden RN, Speight TM, Avery GS . Dantrolene sodium: a review of its pharmacological properties and therapeutic efficacy in spasticity. Drugs 1977; 13: 3–23.
McCleana G . Pharmacological management of neuropathic pain. CNS Drugs 2003; 17: 1031–1043.
Backonja MM . Use of anticonvulsants for treatment of neuropathic pain. Neurology 2002; 59: S14–S17.
Spiller HA, Krenzelok EP, Cookson E . Carbamazepine overdose: a prospective study of serum levels and toxicity. J Toxicol Clin Toxicol 1990; 28: 445–458.
Asconape J . Common issues in the use of antiepileptic drugs. Semin Neurol 2002; 22: 27–39.
Mesdjian E et al. Metabolism of carbamazepine by CYP3A6: a model for in vitro drug interactions studies. Life Sci 1999; 64: 827–835.
Dresser GK, Spence JD, Bailey DG . Pharmacokinetic–pharmacodynamic consequences and clinical relevance of cytochrome P450 3A4 inhibition. Clin Pharmacokinet 2000; 38: 41–57.
Pisani F, Perucca E, Di Perri R . Clinically relevant anti-epileptic drug interactions. J Int Med Res 1990; 18: 1–15.
Sproule BA, Naranjo CA, Brenmer KE, Hassan PC . Selective serotonin reuptake inhibitors and CNS drug interactions. A critical review of the evidence. Clin Pharmacokinet 1997; 33: 454–471.
Spina E, Pisani F, Perucca E . Clinically significant pharmacokinetic interactions with Carbamazepine. An update. Clin Pharmacokinet 1996; 31: 198–214.
Yaich M, Popon M, Medard Y, Aigrain EJ . In-vitro cytochrome P450 dependent metabolism of Oxybutynin to N-desethyloxybutynin in humans. Pharmacogenetics 1998; 8: 449–451.
Lukkari E, Taavitsainen P, Juhakoski A, Pelkonen O . Cytochrome P450 specificity of metabolism and interactions of Oxybutynin in human liver microsomes. Pharmacol Toxicol 1998; 82: 161–166.
Grozinger M, Hartter S, Hiemke C, Roschke J . Oxybutynin enhances the metabolism of clomipramine and dextrorphan possibly by induction of cytochrome P450 isoenzyme. J Clin Psychopharmacol 1999; 19: 287–289.
Roy S, Francis FT, Born CK, Hamrick ME . Interaction of dantrolene with the hepatic mixed function oxidase system. Res Comm Chem Pathol Pharmacol 1980; 27: 507–520.
Francis KT, Hamrick ME . Dantrolene inhibition of the hepatic mixed function oxidase system. Res Comm Chem Pathol Pharmacol 1979; 23: 69–80.
Johannessen CU, Johannessen SI . Valproate: past, present and future. CNS Drug Rev 2003; 9: 199–216.
Perucca E . Pharmacological and therapeutic properties of valproate: a summary after 35 years of clinical experience. CNS Drugs 2002; 15: 695–714.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vander, T., Odi, H., Bluvstein, V. et al. Carbamazepine toxicity following Oxybutynin and Dantrolene administration: a case report. Spinal Cord 43, 252–255 (2005). https://doi.org/10.1038/sj.sc.3101689
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101689