Abstract
Study design: Prospective clinical trial.
Objectives: To evaluate the efficacy of a specific protocol for prevention of thrombo-embolic disease occurring during the acute stage of spinal cord lesions, based on the simultaneous use of pharmacological plus mechanical procedures.
Setting: Regional Spinal Unit of Florence, Italy.
Introduction: Deep venous thrombosis (DVT) is a dangerous pathology whose first clinical sign can be represented by unexpected pulmonary embolism (PE). Its incidence in acute spinal cord injured (SCI) patients is reported to range between 9% and 90%. Its prevention represents one of the major challenges for the clinicians involved in the care of such patients.
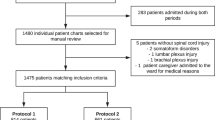
Method: Two hundred and seventy-five SCI patients consecutively admitted to our Centre were investigated by colour doppler ultrasonography of lower limbs and pelvis on admission, after 30–45 days and whenever clinically requested. Subcutaneous Nadroparine, a low molecular weight heparin (LMWH), plus early mobilisation, permanently dressed gradient elastic stockings (PGES), and external sequential pneumatic compression (ESPC) of the lower limbs, applied during the first 30 days after injury, were given to all of them. Colour doppler ultrasonography (CDUS) complete investigations of the lower limbs and pelvis were performed on admission, after 30–45 days and whenever clinically requested. The patients were divided into two groups according to their time interval from injury to the admission to our Centre.
Results: The incidence of detected DVT was 2% in those patients (99) admitted early to our centre (within 72 h from the trauma), who immediately received our prophylactic protocol. No PE was reported. The other group of patients (176), all admitted between 8 and 28 days (mean 12 days) developed DVT in 26% of cases. None of these patients received ESPC before being admitted to our Centre. No patient had been admitted between 3 and 8 days interval time post injury.
Conclusion: Early application of pharmacological plus mechanical treatment for DVT prevention produces a marked reduction in such complications. It also reduces the risks of morbidity and mortality in our patients, and, not least, reduces the hospitalization costs during the early period of rehabilitation.
Similar content being viewed by others
Introduction
Deep vein thrombosis (DVT) represents one of the most common and dangerous complications during the acute stage of spinal cord injuries (SCI),1,2 reported to occur mainly during the first 2 weeks after injury. It represents the first step towards pulmonary embolism (PE) which is still a leading cause of death.3,4,5
Incidence of DVTs in medical reports are extremely variable, ranging from 9% to 90%.6,7,8,9,10,11,12,13,14,15 Nevertheless, in more recent and widespread studies, it is reported to vary between 10% and 30%.16,17,18,19,20,21
With regard to the diagnosis it has been noted that colour doppler ultra sonography (CDUS) is a more available, more economical, more repeatable and a less invasive method compared to phlebography, and it is considered to be the gold standard diagnostic procedure due to its sensitivity and precision.22,23,25
The high risk of DVT in acute SCI patients is due to the simultaneous presence of the three factors of the Virchow's triad: hypercoagulability, stasis, and intimal injury, as stated by Hachen et al.,24 Myllynen et al.10 and Mammen.33
Different ways of prevention have been proposed, with different results: subcutaneous administration of calcium heparin, low molecular weight heparin (LMWH)27,28,29 and low oral dose of dicoumarol are widely used.30 Anti aggregation agents such as aspirin or dipyridamole did not prove to give satisfactory results, when given alone.11
Permanently dressed gradient elastic stockings (PGES) seem to give good results by increasing venous outflow.32
Electrical stimulation applied to the lower limbs has been reported to give some benefit by achieving active muscular contraction and improving venous flow; an enhancement of systemic fibrinolytic activity was also reported.31
The combination of different prophylactic treatments appear to give better results in preventing DVT during the acute stage of SCI.
A retrospective analysis conducted on 492 patients admitted to our centre from January 1979 till December 1995 showed a DVT incidence in the acute stage of 9% in those admitted between 1979 and 1991 (362 patients), when the investigation was performed by clinical examinations followed by venography when clinical evidence was present. DVT incidence rose to 27% in those patients admitted between 1992 and 1995 (130 patients), when all patients were routinely investigated by CDUS on admission, then 30 to 45 days after the lesion and whenever clinically requested. Prophylaxis was homogeneous for all of the 429 patients. The incidence of PE in the first group was 4.3%, that was fatal in 3.5%. PE mostly occurred in undiagnosed DVT and asymptomatic patients, between 15 and 90 days post injury.
In the second group the incidence of PE was 0.77% (1 patient) with no fatal case. Clinical evidence of DVT was present only in 8% of the total investigated population.
By this experience it would appear that a routine CDUS examination might dramatically reduce the incidence of PE together with its mortality rate, by achieving a very early diagnosis of DVT, before any clinical evidence. On the other hand we realized that the real incidence of DVT in acute SCI was very high, reaching 27%, when prophylaxis was performed by giving subcutaneous calcium heparin plus early mobilization of lower limbs and PGES.
A similar trend of incidence of thrombotic pathology has been reported also by Winemiller in 1999.18 Even in that study the progressive increase in incidence was attributed only to a better capability in diagnosing it. From October 1995 we started to adopt a combined approach to prevent DVT, based on the simultaneous use of low molecular weight heparin and external sequential pneumatic compression applied on both the lower limbs.
Methods
All patients affected by acute SCI consecutively admitted to our Centre between January 1st 1996 and May 15th 2000 (275 patients) were investigated by CDUS of lower limbs and pelvis to detect the presence of DVT. The examinations were performed immediately on admission, after 45 to 60 days and then whenever clinically requested. The prophylactic treatment was based on the combined use of mechanical plus pharmacological agents and it was given to 99 patients within 72 h (EAP=Early Admitted Patients), while the other group of 176 patients (LAP=Late Admitted Patients), did not receive the same treatment from the very early stage, but started it from their date of admission, in a period ranging from 7 days to 28 days post injury (mean 12 days).
Inclusion criteria of patients into this study were the following
-
All acute traumatic SCI patients consecutively admitted to our Centre with motor complete and incomplete lesions (ASIA A to D)
-
Absence of contra-indications to heparin and/or to mechanical prophylaxis
Exclusion criteria
-
Formerly known abnormalities of any coagulation factor
-
Any contra-indications to heparin or to lower limb mechanical compression
Our protocol of DVT prevention was:
-
– PGES
-
– Subcutaneous LMWH (nadroparine) 0.4 ml once a day
-
– ESPC of the lower limbs 3 h per day given in two applications
-
– Early mobilisation of the lower limbs
This complete prophylactic treatment lasted at least 30 days from injury, and it was continued only by the application of LMWH and ESPC for two more months according to the patients' progress.
LAP had access to our Centre in an average period of 12 days post trauma, due to the acute treatment performed elsewhere. These patients received in our Centre either the same prophylactic treatment, if no clot had been detected on admission, or the therapy to counteract the eventually detected thrombus (warfarin alone or in association with caval filter). None of these patients had been receiving ESPC as a prophylactic measure in their previous hospitalisation.
Results
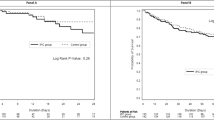
The results are summarised in Tables 1 and 2. DVT incidence in Early Admitted Patients was 2%. In fact only two cases were recorded, both males, one paraplegic ASIA B and the other tetraplegic ASIC C. Those two cases were both detected by CDUS performed on clinical evidence, respectively 25 and 29 days after injury. In one case DVT was just located at popliteal level.
The group of Late Admitted Patients was statistically similar to the group of Early Admitted Patients regarding age, sex, neurological level and ASIA impairment scale and associated injuries. Fisher Exact Test was used for statistical analysis, and results were considered significative when P was 0.1.
The incidence of DVT in Late Admitted Patients was 26%. Of those 60% were detected on admission, while the remaining 40% developed in a period not exceeding 6 weeks of hospitalisation. Sixty-five per cent of detected DVT did not show any evident clinical sign. Asia A were more likely to develop DVT (36%), while in ASIA D on admission only 7% did so. No PE was recorded.
Discussion
A more accurate investigation, such as routine and repeated CDUS in the acute phase, will give a more accurate incidence of DVT. A more accurate investigation may also lead to a more efficient primary prevention of PE, that appears to be a direct consequence of an undiagnosed DVT. In fact, in our study, clinical evidence of DVT was relatively rare, being present in 9% of cases (about 35% of the total DVTs). In other words, the routine investigation by CDUS of lower limbs and pelvis may detect DVT before any clinical evidence, and thus lower the risk of embolism.
Regarding EPSC, the device we have adopted and which has given such positive results is a programmed one, consisting of five contiguous, sequential, pneumatic chambers that cover the foot and the leg. Pneumatic compression is bilaterally applied in sequence, starting from the foot and reaching the proximal area of the lower limb. In every chamber the maximal pressure does not exceed 50 mmHg.
Each compression cycle lasts 90 s with 30 s sequential compression (30 s the first chamber, 24 s the second, 18 s the third and so on) and 60 s deflation time to allow blood flow.
The application of EPSC should not exceed 3 h a day, given in two separate applications daily, in order to avoid further damage to the vessels. Even if it is not statistically correct to compare the two groups of patients for the lack of homogeneity of treatment (the second group had been treated in other hospitals for an average period of 12 days), the important reduction of incidence of DVT using our protocol is considered an important result.
Conclusion
In our study the adoption of a combined approach in DVT prevention that includes LMWH, EPSC and routine plus ‘on demand’ use of CDUS of the lower limbs together with traditional measures (early mobilisation, PGES, clinical and haematological examinations), has dramatically reduced thrombo-embolic disease.
The prophylactic treatment should start during the very early stage post trauma (within 24–72 h) after a requested CDUS examination. We cannot state which one of the two factors of our treatment protocol (LMWH and EPSC) is more important in influencing the results or if the combination of the two is necessary. Further studies will be necessary to give a better understanding of these results. Routine and demand CDUS investigation of lower limbs however revealed this to be an efficacious tool in primary PE prevention.
Finally our study demonstrates the necessity of early admission to Spinal Units for SCI persons to better prevent complications.
References
Walsh JJ, Tribe LR . Phlebothrombosis and pulmonary embolism in paraplegia Paraplegia 1965 3: 209–213
Watson N . Venous thrombosis and pulmonary embolism in spinal cord injury Paraplegia 1968 6: 113–121
Tribe CR . Causes of death in early and late stages of paraplegia Paraplegia 1963 1: 19–47
Stover SL et al.. University of Alabama in Birmingham Spinal Cord Injury Data Management Service. Quarterly Report for the period March 1, 1983 through June 30, 1983 pp 21–22
Winchelli RJ et al. Risk factors associated with pulmonary embolism despite routine prophylaxis: implications for improved protection J Trauma 2: 600–605
Casas ER, Sanchez MP, Arias CR, Masip JP . Prophylaxis of venous thrombosis and pulmonary embolism in patients with acute traumatica spinal cord lesions Paraplegia 1977 15: 209–214
Frisbie JH, Sasahara AA . Low dose heparin prophylaxis for deep venous thrombosis in acute spinal cord injury patients: a controlled study Paraplegia 1981 19: 343–346
Brach BB et al. Venous thrombosis in acute spinal cord paralysis J Trauma 1977 17: 289–292
Rossi E, Green D, Rosen J . Sequential changes in factor VIII and platelets preceding deep vein thrombosis in patients with spinal cord injury Br J Haemat 1980 45: 143–151
Myllynen P et al. Deep vein thrombosis and pulmonary embolism in patients with acute spinal cord injury: A comparison with non-paralysed patients immobilised due to spinal fractures J Trauma 1985 25: 541–543
Merli GJ et al. Aetiology, incidence, and prevention of deep vein thrombosis in acute spinal cord injury Arch Phys Med Rehabil 1993 74: 1199–1205
Gunduz S et al. Deep vein thrombosis in spinal cord injured patients Paraplegia 1993 31: 606–610
Perkash A . Experience with the management of deep vein thrombosis in patients with spinal cord injury. Part II: a critical evaluation of the anticoagulant therapy Paraplegia 1980 18: 2–14
Merli GJ et al. Deep vein thrombosis: prophylaxis in acute spinal cord injured patients Arch Phys Med Rehabil 1988 69: 661–664
Waring WP, Karunas RS . Acute spinal cord injuries and the incidence of clinically occurring thromboembolic disease Paraplegia 1991 29: 8–16
Yelnik A et al. Systematic lower limb phlebography in acute spinal cord injury in 147 patients Paraplegia 1991 29: 253–260
Kulkarni JR, Burt AA, Tromans AT, Constable PDL . Prophylactic low dose heparin anticoagulant therapy in patients with spinal cord injuries: a retrospective study Paraplegia 1992 30: 169–172
Winemiller MH, Stolp-Smith KA, Silverstein MD, Therneau TM . Prevention of venous thromboembolism in patients with spinal cord injury: effects of sequential pneumatic compression and heparin J Spinal Cord Med 1999 22: 182–191
Powell M, Kirschblum S, O'Connor KC . Duplex ultrasound screening for deep vein thrombosis in spinal cord injured patients at rehabilitation admission Arch Phys Med Rehabil 1999 80: 1044–1046
Colachis III SC, Clinchot DM . The association between deep venous thrombosis and heterotopic ossification in patients with acute traumatica spinal cord injury Paraplegia 1994 32: 128–129
Waring WP, Karunas RS . Acute spinal cord injuries and the incidence of clinically occurring thromboembolic disease Paraplegia 1991 29: 8–16
Barrellier MT, Jouen E, Creveuil C . Discordances de résultats entre phlébographie et écho-doppler dans le dépistage de thromboses veineuses asymptomatiques après prothèse totale de hanche. Faux-négatifs de la phlébographie ou faux-positifs de l'écho-Doppler? J Mal Vasc 1998 23: 183–190
Barrellier MT, Lenzin B, Landy S, Le Hello C . Prevalence de la thrombose veineuse phlébographie et écho-doppler dans le dépistage de thromboses veineuses asymptomatiques après prothèse totale de hanche. Faux-negatifs de la phlébographie ou faux-positifs de l'écho-Doppler? J Mal Vasc 1998 23: 183–190
Hachen HJ, Rossier AB, Bouvier CA, Ritschard J . Deficiency within the extrinsic prothrombin activator system in patients with acute spinal cord injury Paraplegia 1974 12: 132–138
Barrellier MT, Lenzin B, Landy S, Le Hello C . Prevalance de la thrombose veineuse diagnostique par echographie-doppler des membres inferieurs dans la suspicion d'embolie pulmonaire et dans l'embolie pulmonaire confirmee J Mal Vasc 2001 26: 23–30
Green DM et al. Fixed versus adjusted dose heparin in the prophylaxis of thromboembolism in spinal cord injury J Am Med Ass 1988 260: 1255–1258
Harris S, Chen D, Green D . Enoxaparin for thromboembolism prophylaxis in spinal injury: preliminary report on experience with 105 patients Am J Phys Med Rehabil 1996 75: 326–327
Knudson MM, Morabito D, Paiement GD, Shakleford S . Use of low molecular weight heparin in preventing thromboembolism in trauma patients J Trauma 1996 41: 446–459
Green D et al. Prevention of thromboembolism in spinal cord injury: role of low molecular weight heparin Arch Phys Med Rehabil 1994 75: 290–292
El Masri WS, Silver JR . Prophylactic anticoagulant therapy in patients with spinal cord injury Paraplegia 1981 19: 334–342
Katz RT, Green D, Sullivan T, Yarkony G . Functional electric stimulation to enhance systemic fibrinolytic activity in spinal cord injury patients Arch Phys Med Rehabil 1987 68: 423–426
Wells PA, Lensing, Hirsh J . Graduated compression stockings in the prevention of postoperative venous thromboembolism: a meta-analysis Arch Int Med 1994 154: 67–72
Mammen EF . Pathogenesis of venous thrombosis Chest 1992 102: (6 suppl) 640S–644S
Acknowledgements
This study was not sponsored or assisted by any company, and there is no conflict of interest.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aito, S., Pieri, A., D'Andrea, M. et al. Primary prevention of deep venous thrombosis and pulmonary embolism in acute spinal cord injured patients. Spinal Cord 40, 300–303 (2002). https://doi.org/10.1038/sj.sc.3101298
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101298
Keywords
This article is cited by
-
Perioperatives Management nach traumatischer Querschnittslähmung
Die Unfallchirurgie (2023)
-
Saudi Critical Care Society clinical practice guidelines on the prevention of venous thromboembolism in adults with trauma: reviewed for evidence-based integrity and endorsed by the Scandinavian Society of Anaesthesiology and Intensive Care Medicine
Annals of Intensive Care (2023)
-
A nested case–control study of risk for pulmonary embolism in the general trauma population using nationwide trauma registry data in Japan
Scientific Reports (2021)
-
Asymptomatic tachycardia and acute pulmonary embolism in a case of tuberculosis spondylodiscitis
Spinal Cord Series and Cases (2018)
-
Re: Incidence of pulmonary embolism after the first 3 months of spinal cord injury
Spinal Cord (2016)