Abstract
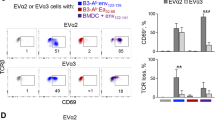
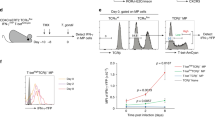
To generate an immune response, antigen-specific T-helper and T-killer cells must find each other and, because they cannot detect each other's presence, they are brought together by an antigen-loaded dendritic cell that displays antigens to both1,2,3. This three-cell interaction, however, seems nearly impossible because all three cell types are rare and migratory. Here we provide a potential solution to this conundrum. We found that the three cells need not meet simultaneously but that the helper cell can first engage and ‘condition’ the dendritic cell, which then becomes empowered to stimulate a killer cell. The first step (help) can be bypassed by modulation of the surface molecule CD40, or by viral infection of dendritic cells. These results may explain the longstanding paradoxical observation that responses to some viruses are helper-independent, and they evoke the possibility that dendritic cells may take on different functions in response to different conditioning signals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Keene, J. A. & Forman, J. Helper activity is required for the in vivo generation of cytotoxic T lymphocytes. J. Exp. Med. 155, 768–782 (1982).
Mitchison, N. A. & O'Malley, C. Three-cell-type clusters of T cells with antigen-presenting cells best explain the epitope linkage and noncognate requirements of the in vivo cytolytic response. Eur. J. Immunol. 17, 1579–1583 (1987).
Bennett, S. R., Carbone, F. R., Karamalis, F., Miller, J. F. & Heath, W. R. Induction of a CD8 cytotoxic T lymphocyte response by cross-priming requires cognate CD4 T cell help. J. Exp. Med. 186, 65–70 (1997).
Guerder, S. & Matzinger, P. Activation versus tolerance: a decision made by helper T cells. Cold Spring Harb. Symp. Quant. Biol. 54, 799 (1989).
Rees, M. A., Rosenberg, A. S., Munitz, T. I. & Singer, A. In vivo induction of antigen-specific transplantation tolerance to Qa1a by exposure to alloantigen in the absence of T-cell help. Proc. Natl Acad. Sci. USA 87, 2765–2769 (1990).
Guerder, S. & Matzinger, P. Afail-safe mechanism for maintaining self-tolerance. J. Exp. Med. 176, 553–564 (1992).
Simpson, E. & Gordon, R. D. Responsiveness to H-Y antigen: Ir gene complementation and target cell specificity. Immunol. Rev. 35, 59–75 (1977).
Gray, D. & Matzinger, P. Tcell memory is short-lived in the absence of antigen. J. Exp. Med. 174, 969–974 (1991).
Fuchs, E. J. & Matzinger, P. Bcells turn off virgin but not memory T cells. Science 258, 1156–1159 (1992).
Ridge, J. P., Fuchs, E. J. & Matzinger, P. Neonatal tolerance revisited: turning on newborn T cells with dendritic cells [see comments]. Science 271, 1723–1726 (1996).
Boog, C. J. et al. Abolition of specific immune response defect by immunization with dendritic cells. Nature 318, 59–62 (1985).
Saeland, S., Duvert, V., Moreau, I. & Banchereau, J. Human B cell precursors proliferate and express CD23 after CD40 ligation. J. Exp. Med. 178, 113–120 (1993).
Cella, M. et al. Ligation of CD40 on dendritic cells triggers production of high levels of interleukin-12 and enhances T cell stimulatory capacity: T-T help via APC activation. J. Exp. Med. 184, 747–752 (1996).
Yang, Y. & Wilson, J. M. CD40 ligand-dependent T cell activation: requirement of B7-CD28 signalling through CD40. Science 273, 1862–1864 (1996).
Bendelac, A. et al. Mouse CD1-specific NK1 T cells: development, specificity, and function. Annu. Rev. Immunol. 15, 535–562 (1997).
Buller, R. M., Holmes, K. L., Hugin, A., Frederickson, T. N. & Morse III, H. C. Induction of cytotoxic T-cell responses in vivo in the absence of CD4 helper cells. Nature 328, 77–79 (1987).
Hou, S., Mo, X. Y., Hyland, L. & Doherty, P. C. Host response to Sendai virus in mice lacking class II major histocompatibility complex glycoproteins. J. Virol. 69, 1429–1434 (1995).
Tripp, R. A., Sarawar, S. R. & Doherty, P. C. Characteristics of the influenza virus-specific CD8 T cell responses in mice homozygous for disruption of the H-2IAb gene. J. Immunol. 155, 2955–2959 (1995).
Ahmed, R., Butler, L. D. & Bhatti, L. T4 T helper cell function in vivo: differential requirement for induction of antiviral cytotoxic T cell and antibody responses. J. Virol. 62, 2102–2106 (1988).
Yong, J. L. et al. Memory B cells from human tonsils colonize mucosal epithelium and directly present antigen to T cells by rapid up-regulation of B7-1 and B7-2. Immunity 2, 239–248 (1995).
Wu, Y. & Liu, Y. Viral induction of co-stimulatory activity on antigen-presenting cells bypasses the need for CD4+ T-cell help in CD8+ T-cell responses. Curr. Biol. 4, 499–505 (1994).
Paliard, X. et al. Simultaneous production of IL-2, IL-4, and IFN-gamma by activated human CD4 and CD8 T cell clones. J. Immunol. 141, 849–855 (1988).
Matzinger, P. Tolerance, danger, and the extended family. Annu. Rev. Immunol. 12, 991–1045 (1994).
Katona, I. M., Urban, J. F. J, Kang, S. S., Paul, W. E. & Finkelman, F. D. IL-4 requirements for the generation of secondary in vivo IgE responses. J. Immunol. 146, 4215–4221 (1991).
Chen, Y., Kuchroo, V. K., Inobe, J., Hafler, D. A. & Weiner, H. L. Regulatory T cell clones induced by oral tolerance: suppression of autoimmune encephalomyelitis. Science 265, 1237–1240 (1994).
Stavnezer, J. Regulation of antibody production and class switching by TGF-beta. J. Immunol. 155, 1647–1651 (1995).
Liu, L., Rich, B. E., Inobe, J., Chen, W. & Weiner, H. L. Apotential pathway of Th2 development during the primary immune response: IL-10 pretreated dendritic cells prime naive CD4+ T cells to secrete IL-4. Adv. Exp. Med. Biol. 417, 375–381 (1997).
Wilbanks, G. A. & Streilein, J. W. Fluids from immune privileged sites endow macrophages with the capacity to induce antigen-specific immune deviation via a mechanism involving transforming growth factor-beta. Eur. J. Immunol. 22, 1031–1036 (1992).
Janeway, C. A. J Approaching the asymptote? Evolution and revolution in immunology. Cold. Spring Harb. Symp. Quant. Biol. 54, 1–13 (1989).
Nonacs, R., Humborg, C., Tam, J. P. & Steinman, R. M. Mechanisms of mouse spleen dendritic cell function in the generation of influenza-specific, cytolytic T lymphocytes. J. Exp. Med. 176, 519–529 (1997).
Acknowledgements
We thank C. Anderson for skin grafting; O. Lantz for the use of his unpublished TCR trangenic mouse; H. Arnheiter, A. Bendelac, L. D'Adamio, R. Germain, R. Schwartz and members of the Ghost lab (O. Alpan, C. Anderson, S. Celli, A. Frank, S. Galluci, T. Kamala, R. Weiss) for reading the manuscript and for useful suggestions; and D. Faherty, K. Hathcock and P. Linsley for blocking reagents.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ridge, J., Di Rosa, F. & Matzinger, P. A conditioned dendritic cell can be a temporal bridge between a CD4+ T-helper and a T-killer cell. Nature 393, 474–478 (1998). https://doi.org/10.1038/30989
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/30989
This article is cited by
-
Immunity to Cryptosporidium: insights into principles of enteric responses to infection
Nature Reviews Immunology (2024)
-
A hepatic network of dendritic cells mediates CD4 T cell help outside lymphoid organs
Nature Communications (2024)
-
CD40 ligand stimulation affects the number and memory phenotypes of human peripheral CD8+ T cells
BMC Immunology (2023)
-
Signaling pathways involved in the biological functions of dendritic cells and their implications for disease treatment
Molecular Biomedicine (2023)
-
Tumor resident memory CD8 T cells and concomitant tumor immunity develop independently of CD4 help
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.