Abstract
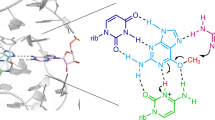
RNase T1 is folded into an α-helix of 4.5 turns, covered by a four-strand antiparallel β-sheet. Specific recognition of 2′-guanylic acid arises from hydrogen bonding between main chain peptide groups and the O-6 and N-1–H of guanine, as well as from stacking of Tyr 45 on guanine. At the active site, Glu 58, His 92 and Arg 77 are involved in phosphodiester hydrolysis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kartha, G. et al. Nature 213, 862–865 (1967).
Carlisle, C. H. et al. J. molec. biol. 85, 1–18 (1974).
Wlodawer, A. et al. J. biol. Chem. 257, 1325–1332 (1982).
Wyckoff, H. W. et al. J. biol. Chem. 245, 305–328 (1970).
Arnone, A. et al. J. biol. Chem. 246, 2302–2316 (1971).
Egami, F. et al. Molec. Biol. Biochem. Biophys. 32, 250–277 (1980).
Takahashi, K. et al. J. biol. Chem. 242, 4682–4690 (1967).
Takahashi, K. J. Biochem., Tokyo 67, 833–839 (1970).
Takahashi, K. J. Biochem., Tokyo 80, 1267–1275 (1976).
Fülling, R. & Rüterjans, H. FEBS Lett. 88, 279–282 (1978).
Arata, Y. et al. Biochemistry 18, 18–24 (1979).
Osterman, H. L. & Walz, F. G. Jr Biochemistry 18, 1984–1988 (1979).
Takahashi, K. J. Biochem., Tokyo 68, 659–664 (1970).
Heinemann, U. et al. Eur. J. Biochem. 109, 109–114 (1980).
Stout, G. H. & Jensen, L. H. X-Ray Structure Determination (Collier-Macmillan, London, 1968).
North, A. C. T. et al. Acta crystallogr. A 24, 351–359 (1968).
Matthews, B. W. Acta crystallogr. 20, 230–239 (1966).
Main, P. et al. MULTAN. A System of Computer Programs for the Automatic Solution of Crystal Structures from X-ray Diffraction Data (University of York, UK and University of Louvain, Belgium, 1977).
Blow, D. M. & Crick, F. H. C. Acta crystallogr. 12, 794–802 (1959).
Richards, F. M. J. molec. Biol. 37, 225–230 (1968).
Takahashi, K. J. Biochem., Tokyo 70, 945–960 (1971).
Richardson, J. S. et al. Proc. natn. Acad. Sci. U.S.A. 75, 2574–2578 (1978).
Irie, M. J. Biochem., Tokyo 63, 649–653 (1968).
Osterman, H. L. & Walz, F. G. Jr Biochemistry 17, 4124–4130 (1978).
Kanaya, S. & Uchida, T. J. Biochem., Tokyo 89, 591–597 (1981).
Yamamoto, Y. et al. Nucleic Acids Res. Symp. Ser. 10, 227–231 (1981).
Hartley, R. W. J. molec. Evol. 15, 355–358 (1980).
Takahashi, K. J. Biochem., Tokyo 72, 1469–1482 (1972).
Oshima, T. & Imahori, K. J. Biochem., Tokyo 70, 197–199 (1971).
Guschlbauer, W. & Courtois, Y. FEBS Lett. 1, 183–186 (1968).
Son, T.-D. et al. J. Am. chem. Soc. 94, 7903–7911 (1972).
Kyogoku, Y. et al. J. Biochem., Tokyo 91, 675–679 (1982).
Pongs, O. Biochemistry 9, 2316–2321 (1970).
Seeman, N. C. et al. Proc. natn. Acad. Sci. U.S.A. 73, 804–808 (1976).
Helene, C. & Lancelot, G. Prog. Biophys. molec. Biol. 39, 1–68 (1982).
Rüterjans, A. & Pongs, O. Eur. J. Biochem. 18, 313–318 (1971).
Inagaki, F. et al. J. Biochem., Tokyo 89, 1185–1195 (1981).
Kimura, S. et al. J. Biochem., Tokyo 85, 301–310 (1979).
Iida, S. & Ooi, T. Biochemistry 8, 3897–3901 (1969).
Usher, D. A. Proc. natn. Acad. Sci. U.S.A. 62, 661–667 (1969).
Eckstein, F. et al. Biochemistry 11, 3507–3512 (1972).
Richards, F. M. & Wyckoff, H. W. The Enzymes Vol. 4 (ed. Boyer, P. D.) 647–806 (Academic, New York, 1971).
Wodak, S. Y. et al. J. molec. Biol. 116, 855–875 (1977).
Cotton, F. A. et al. Proc. natn. Acad. Sci. U.S.A. 76, 2551–2555 (1979).
Mauguen, Y. et al. Nature 297, 162–164 (1982).
Ohgi, K. et al. J. Biochem., Tokyo 90, 113–123 (1981).
Ikehara, M. & Imura, J. Chem. pharm. Bull. 29, 2408–2412 (1981).
Fülling, R. thesis, Univ. Münster (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Heinemann, U., Saenger, W. Specific protein-nucleic acid recognition in ribonuclease T1–2′-guanylic acid complex: an X-ray study. Nature 299, 27–31 (1982). https://doi.org/10.1038/299027a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/299027a0
This article is cited by
-
Hydrolysis of a slow cyclic thiophosphate substrate of RNase T1 analyzed by time-resolved crystallograph
Nature Structural Biology (1998)
-
An engineered ribonuclease preferring phosphorothioate RNA
Nature Structural Biology (1998)
-
Tryptophan phosphorescence of ribonuclease T1 as a probe of protein flexibility
Journal of Fluorescence (1992)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.