Abstract
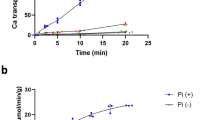
Primary active transport systems establish electrochemical gradients of their transport substrates at the direct expense of chemical or light energy. The chemical energy can be provided by cleavage of the phosphoric anhydride bond of ATP, establishing Na+, K+, H+ or Ca2+ gradients with the respective ATPases. Whereas in eukaryotic cells the Na+ and K+ imbalance between cells and the surrounding fluids is brought about by the (Na+ + K+)ATPase1, no such enzyme is known for bacterial cells. Generally, the gradients of Na+ and K+ ions in these cells are believed to be generated by secondary transport systems consuming the energy provided by the electrochemical gradient of protons2,3. In some bacterial species, however, sodium ions may be transported by primary ATP or light-driven sodium pumps4,5. The primary sodium pump oxaloacetate decarboxylase represents a novel energy conversion mechanism in that it consumes the chemical energy of the decarboxylation of oxaloacetate to drive Na+ transport6–8. We show here that methylmalonyl-CoA decarboxylase is another example of a Na+ transport enzyme converting the chemical energy of a decarboxylation reaction into an ion gradient.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Glynn, I. M. & Karlish, S. J. D. A. Rev. Physiol. 37, 13–55 (1975).
Mitchell, P. J. Bioenerg. 4, 53–91 (1973).
Rosen, B. P. & Kashket, E. R. in Bacterial Transport (ed. Rosen, B.) 559–620 (Dekker, New York, 1978).
Heefner, D. L., Kobayashi, H. & Harold, F. M. J. biol. Chem. 255, 11403–11407 (1980).
McDonald, R. E., Greene, R. V., Clark, R. D. & Lindley, E. V. J. biol Chem. 254, 11831–11838 (1979).
Dimroth, P. FEBS Lett. 122, 234–236 (1980).
Dimroth, P. Eur. J. Biochem. 121, 443–449 (1982).
Dimroth, P. J. biol. Chem. 256, 11974–11976 (1981).
Stern, J. R. Biochemistry 6, 3545–3551 (1967).
Dimroth, P. Eur. J. Biochem. 115, 353–358 (1981).
Dimroth, P. Eur. J. Biochem. 121, 435–441 (1982).
Galivan, J. H. & Allen, S. H. G. J. biol. Chem. 243, 1253–1261 (1968).
Galivan, J. H. & Allen, S. H. G. Archs Biochem. Biophys. 126, 838–847 (1968).
Henrikson, K. P., Allen, S. H. G. & Maloy, W. L. Analyt. Biochem. 94, 366–370 (1979).
Racker, E., Violand, B., O'Neal, S., Alfonzo, M. & Telford, J. Archs Biochem. Biophys. 198, 470–477 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hilpert, W., Dimroth, P. Conversion of the chemical energy of methylmalonyl-CoA decarboxylation into a Na+gradient. Nature 296, 584–585 (1982). https://doi.org/10.1038/296584a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/296584a0
This article is cited by
-
Effects of adding eubiotic lignocellulose on the performance, the gut microbiota, and short-chain fatty acids of layer chickens
Brazilian Journal of Microbiology (2022)
-
Metabolic pathway to propionate of Pectinatus frisingensis, a strictly anaerobic beer-spoilage bacterium
Archives of Microbiology (1994)
-
Anaerobic degradation of 4-hydroxybenzoate: Reductive dehydroxylation of 4-hydroxybenzoyl-CoA and ATP formation during 4-hydroxybenzoate decarboxylation by the phenol-metabolizing bacteria of a stable, strictly anaerobic consortium
Applied Microbiology and Biotechnology (1994)
-
Growth yield increase and ATP formation linked to succinate decarboxylation in Veillonella parvula
Archives of Microbiology (1992)
-
Propionate acts as carboxylic group acceptor in aspartate fermentation by Propionibacterium freudenreichii
Archives of Microbiology (1990)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.