Abstract
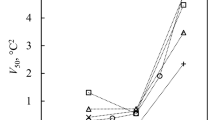
The process by which ice forms during the cooling of water and aqueous solutions has been studied intensively mostly in conditions where the nucleation occurs heterogeneously—on some particulate impurity surface. When the more fundamental homogeneous process has been deliberately investigated1–3 it has nearly always been in conditions in which the crystallization process, once initiated, occurs with great rapidity1,2. Seeking an additional ‘degree of freedom’ in the investigation of this important phenomenon, we have initiated the crystallization in conditions where the growth is extremely slow and the nucleation events can be controlled. Our observations are described below.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wood, G. R. & Walton, A. G. J. appl. Phys. 41, 3027 (1970).
Rasmussen, D. H. & MacKenzie, A. P. Water Structure at the Water Polymer Interface, (ed. Jellinek, H. H. G.) 126–145 (Plenum, New York, 1972).
Kanno, H. & Angell, C. A. J. phys. Chem. 81, 2639 (1977).
Angell, C. A., Sare, E. J., Donnella, J. & MacFarlane, D. R. J. phys. Chem. 85, 1461 (1981).
Wright, A. F., Talbot, J. & Fender, B. E. F. Nature 227, 366 (1979).
Wright, A. F. in Symp. on Neutron Scattering, Argonne (American Institute of Physics Symp. Ser., in the press).
Elarby, A. et al. J. Phys. Lett. (submitted).
Angell, C. A. & MacFarlance, D. R. in Advances in Ceramics (in the press).
Bressel, R. D. thesis, Purdue Univ. (1972).
Hsich, S. Y., Gammon, R. W. & Montrose, C. J. J. chem. Phys. 56, 1666 (1972).
Angell, C. A. & Sare, E. J. J. chem. Phys. 49, 4714 (1968).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dupuy, J., Jal, J., Ferradou, C. et al. Controlled nucleation and quasi-ordered growth of ice crystals from low temperature electrolyte solutions. Nature 296, 138–140 (1982). https://doi.org/10.1038/296138a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/296138a0
This article is cited by
-
Disentangling kinetics from thermodynamics in heterogeneous colloidal systems
Nature Communications (2023)
-
Tude par ACD du domaine hors d'equilibre du systemexLiCl-(1−x)H2O (0
Journal of Thermal Analysis (1984)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.