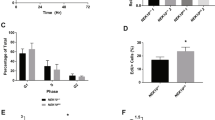
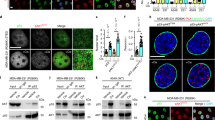
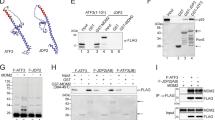
Abstract
The tumour suppressor p53 becomes activated as a transcription factor in response to DNA damage1,2,3, but the mechanism for this activation is unclear. A good candidate for an upstream activator of p53 is the DNA-dependent protein kinase (DNA-PK) that depends on the presence of DNA breaks for its activity4,5,6. Here we investigate the link between DNA damage and the activation of DNA-PK and of p53. To determine whether DNA-PK is an upstream mediator of the p53 DNA-damage response, we analysed a severe combined-immunodeficiency (SCID) mouse cell line, SCGR11 (refs 7, 8), and the human glioma cell line M059J (ref. 9). Both cell lines lack any detectable DNA-PK activity. We find that p53 is incapable of binding to DNA in the absence of DNA-PK, that DNA-PK is necessary but not sufficient for activation of p53 sequence-specific DNA binding, and that this activation occurs in response to DNA damage. Our results establish DNA-PK as a link between DNA damage and p53 activation, and reveal the existence of a mammalian DNA-damage-response pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kastan, M. B., Onyekwere, O., Sidransky, D., Vogelstein, B. & Craig, R. W. Participation of p53 in the cellular response to DNA damage. Cancer Res. 51, 6304–6311 (1991).
Tishler, R. B., Calderwood, S. K., Coleman, C. N. & Price, B. D. Increases in sequence-specific DNA binding by p53 following treatment with chemotherapeutic and DNA damaging agents. Cancer Res. 53, 2212–2216 (1993).
Nelson, W. G. & Kastan, M. B. DNA strand breaks: The DNA template alterations that trigger p53-dependent DNA damage response pathways. Mol. Cell. Biol. 14, 1815–1823 (1994).
Anderson, C. W. & Lees-Miller, S. P. The human DNA-activated protein kinase DNA-PK. Crit. Rev. Eukaryot. Gene Expr. 2, 283–314 (1992).
Gottlieb, T. M. & Jackson, S. P. The DNA-dependent protein kinase: requirement for DNA ends and association with Ku antigen. Cell 72, 131–142 (1993).
Morozov, V. E., Falzon, M., Anderson, C. W. & Kuff, E. L. DNA-dependent protein kinase is activated by nicks and larger single-stranded gaps. J. Biol. Chem. 269, 16684–16688 (1994).
Hendrickson, E. A. et al. Alink between double-strand break-related repair and V(D)J recombination: The SCID mutation. Proc. Natl Acad. Sci. USA 88, 4061–4065 (1991).
Blunt, T. et al. Defective DNA-dependent protein kinase activity is linked to V(D)J recombination and DNA repair defects associated with the murine SCID mutation. Cell 80, 813–823 (1995).
Lees-Miller, S. P. et al. Absence of p350 subunit of DNA-activated protein kinase from a radiosensitive human cell line. Science 267, 1183–1185 (1995).
Kern, S. E. et al. Oncogenic forms of p53 inhibit p53-regulated gene expression. Science 256, 827–830 (1992).
Harper, J. W., Adami, G. R., Wei, N., Keyomarsi, K. & Elledge, S. J. The p21 Cdk-interacting protein Cip1 is a potent inhibitor of G1 cyclin-dependent kinases. Cell 75, 805–816 (1993).
El-Diery, W. S. et al. WAF1, a potential mediator of p53 tumor suppression. Cell 75, 817–825 (1993).
Bogue, M. A., Zhu, C., Aguilar-Cordova, E., Donehower, L. A. & Both, D. B. p53 is required for both radiation-induced differentiation and rescue of V(D)J rearrangement in SCID mouse thymocytes. Genes Dev. 10, 553–565 (1996).
Guidos, C. J. et al. V(D)J recombination activates a p53-dependent DNA damage checkpoint in SCID lymphocyte precursors. Genes Dev. 10, 2038–2054 (1996).
Huang, L., Clarkin, K. C. & Wahl, G. M. p53-dependent cell cycle arrests are preserved in DNA-activated protein kinase-deficient mouse fibroblasts. Cancer Res. 56, 2940–2944 (1996).
Nacht, M. et al. Mutations in the p53 and SCID genes cooperate in tumorigenesis. Genes Dev. 10, 2055–2066 (1996).
Zhu, C., Bogue, M. A., Lim, D., Hasty, P. & Roth, D. B. Ku86-deficient mice exhibit severe combined immunodeficiency and defective processing of V(D)J recombination intermediates. Cell 86, 379–389 (1996).
Jhappan, C., Morse, H. C., Fleischmann, R. D., Gottesman, M. M. & Merlino, G. DNA-PKCS: a T-cell tumour supressor encoded at the mouse scid locus. Nature Genet. 17, 483–486 (1997).
Hollstein, M. et al. Database of p53 gene somatic mutations in human tumors and cell lines. Nucleic Acids Res. 22, 3551–3555 (1994).
Lees-Miller, S. P., Sakaguchi, K., Ullrich, S., Appella, E. & Anderson, C. W. Human DNA-activated protein kinase phosphorylates serines 15 and 37 in the amino-terminal transactivation domain of human p53. Mol. Cell. Biol. 12, 5041–5049 (1992).
Shieh, S.-Y., Ikeda, M., Taya, Y. & Prives, C. DNA damage-induced phosphorylation of p53 alleviates inhibition by MDM2. Cell 91, 325–334 (1997)
Abbondanzo, S. J., Gadi, I. & Stewart, C. L. Derivation of embryonic stem cell lines. Methods Enzymol. 225, 803–823 (1993).
McLure, K. G. & Lee, P. W. K. How p53 binds DNA as a tetramer. EMBO J. 17, 3342–3350 (1998).
Chen, C. & Okayama, H. High-efficiency transformation of mammalian cells by plasmid DNA. Mol. Cell. Biol. 7, 2745–2752 (1987).
Neumann, J. R., Morency, C. A. & Russian, K. O. Anovel rapid assay for chloramphenicol acetyltransferase gene expression. Biotechniques 5, 444–448 (1987).
Wong, H., Anderson, W. D., Cheng, T. & Riabowol, K. T. Monitoring mRNA expression by polymerase chain reaction: The “primer-dropping” method. Anal. Biochem. 223, 251–258 (1994).
Skup, D. & Millward, S. Reovirus-induced modification of cap-dependent translation in infected L cells. Proc. Natl Acad. Sci. USA 77, 152–156 (1980).
Milner, J., Medcalf, E. A. & Cook, A. C. Tumor suppressor p53: Analysis of wild-type and mutant p53 complexes. Mol. Cell. Biol. 11, 12–19 (1991).
Acknowledgements
We thank P. Jeggo for SCGR11 SCID cells, J. Allalunis-Turner for M059J and M059K cells, J. Milner for the wild-type murine p53 plasmid pSP6p53-Ala, B. Vogelstein for pG13CAT, J. Martinez for the pAb421 antibody, D. Chan for purifying DNA-PK, and E. Rattner and B. Carson for help with the preparation of mouse embryo fibroblasts. This work was supported by a grant (to P.W.K.L.) from the Alberta Cancer Board; K.G.M. was a recipient of the Alberta Heritage Foundation for Medical Research studentship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Woo, R., McLure, K., Lees-Miller, S. et al. DNA-dependent protein kinase acts upstream of p53 in response to DNA damage. Nature 394, 700–704 (1998). https://doi.org/10.1038/29343
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/29343
This article is cited by
-
Role of PRKDC in cancer initiation, progression, and treatment
Cancer Cell International (2021)
-
Genotoxic stress-activated DNA-PK-p53 cascade and autophagy cooperatively induce ciliogenesis to maintain the DNA damage response
Cell Death & Differentiation (2021)
-
Human p53 interacts with the elongating RNAPII complex and is required for the release of actinomycin D induced transcription blockage
Scientific Reports (2017)
-
DNA repair kinetics in SCID mice Sertoli cells and DNA-PKcs-deficient mouse embryonic fibroblasts
Chromosoma (2017)
-
β-Carotene-induced apoptosis is mediated with loss of Ku proteins in gastric cancer AGS cells
Genes & Nutrition (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.