Abstract
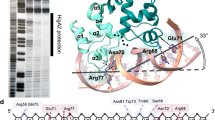
The virulent phenotype of the pathogenic bacterium Corynebacterium diphtheriae is conferred by diphtheria toxin, whose expression is an adaptive response to low concentrations of iron. The expression of the toxin gene (tox) is regulated by the repressor DtxR (ref. 1), which is activated by transition metal ions. X-ray crystal structures of DtxR with2,3,4,5 and without (apo-form2) its coordinated transition metal ion have established the general architecture of the repressor, identified the location of the metal-binding sites, and revealed a metal-ion-triggered subunit–subunit ‘caliper-like’ conformational change. Here we report thethree-dimensional crystal structure of the complex between a biologically active Ni(ii)-bound DtxR(C102D) mutant, in whicha cysteine is replaced by an aspartate at residue 102, and a 33-base-pair DNA segment containing the toxin operator tox O. This structure shows that DNA interacts with two dimeric repressor proteins bound to opposite sides of the tox operator. We propose that a metal-ion-induced helix-to-coil structural transition in the amino-terminal region of the protein is partly responsible for the unique mode of repressor activation by transition metal ions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Tao, X., Schiering, N., Zeng, H. Y., Ringe, D. & Murphy, J. R. Iron, DtxR, and the regulation of diphtheria toxin expression. Mol. Microbiol. 14, 191–197 (1994).
Schiering, N.et al. Structures of the apo- and the metal ion-activated forms of the diphtheria tox repressor from Corynebacterium diphtheriae.. Proc. Natl Acad. Sci. USA 92, 9843–9850 (1995).
Ding, X., Zeng, H., Schiering, N., Ringe, D. & Murphy, J. R. Identification of the primary metal ion-activation sites of the diphtheria tox repressor by X-ray crystallography and site-directed mutational analysis. Nature Struct. Biol. 3, 382–387 (1996).
Qiu, X.et al. Three-dimensional structure of the diphtheria toxin repressor in complex with divalent cation co-repressors. Structure 3, 87–100 (1995).
Qiu, X., Pohl, E., Holmes, R. K. & Hol, W. G. High-resolution structure of the diphtheria toxin repressor complexed with cobalt and manganese reveals an SH3-like third domain and suggests a possible role of phosphate as a co-repressor. Biochemsitry 35, 12292–12302 (1996).
Somers, W. S. & Phillips, S. E. V. Crystal structure of the met repressor–operator complex at 2.8 Å resolution reveals DNA recognition by β-strands. Nature 359, 387–393 (1992).
Tao, X. & Murphy, J. R. Binding of the metalloregulatory protein DtxR to the diphtheria tox operator requires a divalent heavy metal ion and protects the palindromic sequence from DNase I digestion. J. Biol. Chem. 267, 21761–21764 (1992).
Must, L. M., Twiddy, E. M., Sabol, S. Z. & Holmes, R. K. in Bacterial Protein Toxins Vol. 6(eds Hacker, J., Rappuoli, R., Alouf, J., Fehernbach, F. & Feer, J.) (Fisher Stuttgart, Germany, in the press).
Tao, X. & Murphy, R. R. Cysteine-102 is positioned in the metal binding activation site of the Corynebacterium diphtheriae regulatory element DtxR. Proc. Natl Acad. Sci. USA 90, 8524–8528 (1993).
Kercher, M. A., Lu, P. & Lewis, M. Lac repressor–operator complex. Curr. Opin. Struct. Biol. 7, 76–85 (1997).
Albright, R. A. & Matthews, B. W. How Cro and λ-repressor distinguish between operators: The structural basis underlying a genetic switch. Proc. Natl Acad. Sci. USA 95, 3431–3436 (1998).
Schmitt, M. P. & Holmes, R. K. Analysis of diphtheria toxin repressor-operator interactions and characterization of a mutant repressor with decreased binding activity for divalent metals. Mol. Microbiol. 9, 173–181 (1993).
Wang, Z., Schmitt, M. P. & Holmes, R. K. Characterization of mutations that inactivate the diphtheria toxin repressor gene (dtxR). Infect. Immun. 62, 1600–1608 (1994).
Tao, X., Zeng, H. Y. & Murphy, J. R. Transition metal ion activation of DNA binding by the diphtheria tox repressor requires the formation of stable homodimers. Proc. Natl Acad. Sci. USA 92, 6803–6807 (1995).
Otwinowski, Z.et al. Crystal structure of trp repressor/operator complex at atomic resolution. Nature 335, 321–329 (1988).
Lawson, C. L. & Carey, J. Tandem binding in crystals of a trp repressor/operator half-site complex. Nature 366, 178–182 (1993).
Zhang, H.et al. The solution structure of the trp repressor-operator DNA complex. J. Mol. Biol. 238, 592–614 (1994).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Meth. Enzymol. 276, 307–326 (1997).
Navaza, J. AMoRe: an automated package for molecular replacement. Acta Crystallogr. A 50, 157–163 (1994).
Collaborative Computational Project Number 4. The CCP4 suite: Programs for protein crystallography Version 3.1. Acta Crystallogr. D 50, 760–763 (1994).
Brünger, A. T., Kuriyan, J. & Karplus, M. Crystallographic R -factor refinement by molecular dynamics. Science 235, 458–460 (1987).
Hodel, A., Kim, S.-H. & Brünger, A. T. Model bias in macromolecular crystal structures. Acta Crystallogr. A 48, 851–858 (1992).
Jones, T. A., Zou, J.-Y., Cowan, S. W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Laskowski, R. A., MacArthur, M. W., Moss, D. S. & Thornton, J. M. PROCHECK: a program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 26, 283–291 (1993).
Engh, R. A. & Huber, R. Accurate bond and angle parameters for X-ray protein structure refinement. Acta Crystallogr. A 47, 392–400 (1991).
Brünger, A. T. Free R value: a novel statistical quantity for assessing the accuracy of crystal structures. Nature 355, 472–475 (1992).
Boyd, J., Oza, M. N. & Murphy, J. R. Molecular cloning and DNA sequence analysis of a diphtheria tox iron-dependent regulatory element (dtxR) from Corynebacterium diphtheriae. Proc. Natl Acad. Sci. USA 87, 5968–5972 (1990).
Ravishankar, G., Swaminathan, S., Beveridge, D. L., Lavery, R. & Sklenar, H. Conformational and helical analysis of 30ps of molecular dynamics on the d(CGCGAATTCGCG) double helix: ‘Curves’, dials and windows. J. Biomol. Struct. Dynam. 6, 669–699 (1989).
Kraulis, P. J. MOLSCRIPT: A program package to produce both detailed and schematic plots of protein structures. J. Appl. Crystallogr. 24, 946–950 (1991).
Read, R. J. Improved fourier coefficients for maps using phases from partial structures with errors. Acta Crystallogr. A 42, 140–149 (1986).
Acknowledgements
This work was supported by a Public Health Service Grant from the National Institute of Allergy and Infectious Diseases and (in part) by a grant from the Lucille P. Markey Charitable Trust. We thank M. Stanton and Y. Wang for help with data collection, D. Peisach and E. Peisach for rendering images, and N. Schiering, S. Chen and G. A. Petsko for discussion.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
White, A., Ding, X., vanderSpek, J. et al. Structure of the metal-ion-activated diphtheria toxin repressor/ tox operator complex. Nature 394, 502–506 (1998). https://doi.org/10.1038/28893
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/28893
This article is cited by
-
Biochemical characterization of ferric uptake regulator (Fur) from Aliivibrio salmonicida. Mapping the DNA sequence specificity through binding studies and structural modelling
BioMetals (2020)
-
Dynamic equilibrium on DNA defines transcriptional regulation of a multidrug binding transcriptional repressor, LmrR
Scientific Reports (2017)
-
Ferric Uptake Regulator (FUR) protein: properties and implications in cyanobacteria
Annals of Microbiology (2016)
-
Mechanistic insights into metal ion activation and operator recognition by the ferric uptake regulator
Nature Communications (2015)
-
Protein photocrosslinking reveals dimer of dimers formation on MarR protein in Escherichia coli
Science China Chemistry (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.