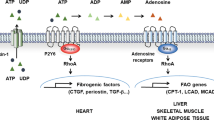
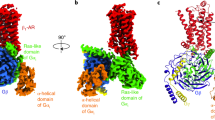
Abstract
How does a trimeric G protein on the inside of a cell membrane respond to activation by a transmembrane receptor? G-protein mutations in patients with hypertension and inherited endocrine disorders enhance or block signals from stimulated receptors. In combination with three-dimensional crystal structures and results from biochemical experiments, the phenotypes produced by these mutations suggest a model for the molecular activation mechanism that relays hormonal and sensory signals transmitted by many transmembrane receptors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hamm, H. E. The many faces of G protein signaling. J. Biol. Chem. 273, 669–672 (1998).
Sprang, S. R. Gprotein mechanisms: Insights from structural analysis. Annu. Rev. Biochem. 66, 639–678 (1997).
Lambright, D. G. et al. The 2.0 Å crystal structure of a heterotrimeric G protein. Nature 379, 311–319 (1996).
Baldwin, J. M., Schertler, G. F. & Unger, V. M. An alpha-carbon template for the transmembrane helices in the rhodopsin family of G-protein-coupled receptors. J. Mol. Biol. 272, 144–164 (1997).
Hargrave, P. A., Hamm, H. E. & Hofmann, K. P. Interaction of rhodopsin with the G-protein, transducin. Bioessays 15, 43–50 (1993).
Fahmy, K., Siebert, F. & Sakmar, T. P. Photoactivated state of rhodopsin and how it can form. Biophys. Chem. 56, 171–181 (1995).
Strader, C. D., Fong, T. M., Graziano, M. P. & Tota, M. R. The family of G-protein-coupled receptors. FASEB J. 9, 745–754 (1995).
Bourne, H. R. How receptors talk to trimeric G proteins. Curr. Opin. Cell Biol. 134–142 (1997).
Bourne, H. R., Sanders, D. A. & McCormick, F. The GTPase superfamily. A conserved switch for diverse cell functions. Nature 348, 125–132 (1990).
Bourne, H. R., Sanders, D. A. & McCormick, F. The GTPase superfamily: Conserved structure and molecular mechanism. Nature 349, 117–127 (1991).
Bourne, H. R. The arginine finger strikes again. Nature 389, 673–674 (1997).
Bohm, A., Gaudet, R. & Sigler, P. B. Structural aspects of heterotrimeric G-protein signaling. Curr. Opin. Biotechnol. 8, 480–487 (1997).
Scheffzek, K. et al. The Ras–RasGAP complex: Structural basis for GTPase activation and its loss in oncogenic ras mutants. Science 277, 333–338 (1997).
Sondek, J., Lambright, D. G., Noel, J. P., Hamm, H. E. & Sigler, P. B. GTPase mechanism of G proteins from the 1.7-Å crystal structure of transducin α-GDP-A1F4−. Nature 372, 276–279 (1994).
Coleman, D. E. et al. Structure of active conformations of Giα1 and the mechanism of GTP hydrolysis. Science 265, 1405–1412 (1994).
Tesmer, J. J., Berman, D. M., Gilman, A. G. & Sprang, S. R. Structure of RGS4 bound to AlF4− activated Giα1: stabilization of the transition state for GTP hydrolysis. Cell 89, 251–261 (1997).
Lambright, D. G., Noel, J. P., Hamm, H. E. & Sigler, P. B. The 1.8 Å crystal structure of transducin α-GDP: structural determinants for activation of a heterotrimeric G-protein α-subunit. Nature 369, 621–628 (1994).
Noel, J. P., Hamm, H. E. & Sigler, P. B. The 2.2 Å crystal structure of transducin α-GTPγS. Nature 366, 654–663 (1993).
Sondek, J., Bohm, A., Lambright, D. G., Hamm, H. E. & Sigler, P. B. Crystal structure of a GAprotein βγ dimer at 2.1 Å resolution. Nature 379, 369–374 (1996).
Mixon, M. B. et al. Tertiary and quaternary structural changes in Giα1induced by GTP hydrolysis. Science 270, 954–960 (1995).
Wall, M. A. et al. The structure of the G protein heterotrimer Giα1β1γ1. Cell 83, 1047–1058 (1995).
Sunahara, R. K., Tesmer, J. J. G., Gilman, A. G. & Sprang, S. R. Crystal structure of the adenylyl cyclase activator, Gsα. Science 278, 1943–1947 (1997).
Garcia, P. D., Onrust, R., Bell, S. M., Sakmar, T. P. & Bourne, H. R. Transducin-α C-terminal mutations prevent activation by rhodopsin: A new assay using recombinant proteins expressed in cultured cells. EMBO J. 14, 4460–4469 (1995).
Onrust, R. et al. Receptor and βγ binding sites in the α subunit of the retinal G protein transducin. Science 275, 381–384 (1997).
Hamm, H. E. et al. Site of G protein binding to rhodopsin mapped with synthetic peptides from the α subunit. Science 241, 832–835 (1988).
Kawashima, T., Berthet-Colominas, C., Wulff, M., Cusack, S. & Leberman, R. The structure of the Escherichia coli EF-Tu·EF-Ts complex at 2.5 Å resolution. Nature 379, 511–518 (1996).
Wang, Y., Jiang, Y., Meyering-Voss, M., Sprinzl, M. & Sigler, P. B. Crystal structure of the EF-Tu·EF-Ts complex from Thermus thermophilus. Nature Struct. Biol. 4, 950–956 (1997).
Iiri, T., Herzmark, P., Nakamoto, J. M., Van Dop, C. & Bourne, H. R. Rapid GDP release from Gsαin patients with gain and loss of endocrine function. Nature 371, 164–168 (1994).
Feig, L. A. & Cooper, G. M. Relationship among guanine nucleotide exchange, GTP hydrolysis, and transofrming potential of mutated ras proteins. Mol. Cell. Biol. 8, 2472–2478 (1988).
Schwindinger, W. F., Miric, A., Zimmerman, D. & Levine, M. A. Anovel Gsα mutant in a patient with Albright hereditrary osteodystrophy uncouples cell surface receptors from adenylyl cyclase. J. Biol. Chem. 269, 25387–25391 (1994).
West, R. E., Moss, J., Vaughan, M., Liu, T. & Liu, T. Y. Pertussis toxin-catalyzed ADP-ribosylation of transducin. Cysteine-347 is the ADP-ribose acceptor site. J. Biol. Chem. 260, 14428–14430 (1985).
Farrens, D. L., Altenbach, C., Yang, K., Hubbell, W. L. & Khorana, . G. Requirement of rigid-body motion of transmembrane helices for light activation of rhodopsin. Science 274, 768–770 (1996).
Bae, H. et al. Molecular determinants of selectivity in 5-hydroxytryptamine1Breceptor–G protein interactions. J. Biol. Chem. 272, 32071–32077 (1997).
Mazzoni, M. R. & Hamm, H. E. Interaction of transducin with light-activated rhodopsin protects It from proteolytic digestion by trypsin. J. Biol. Chem. 271, 30034–30040 (1996).
Denker, B. M., Schmidt, C. J. & Neer, E. J. Promotion of the GTP-liganded state of the Goαprotein by deletion of the C terminus. J. Biol. Chem. 267, 9998–10002 (1992).
Ford, C. E. et al. Molecular basis for interactions of G protein βγ subunits with effectors. Science 280, 1271–1274 (1998).
Siffert, W. et al. Association of an allele of a human G-protein β3 subunit gene with hypertension. Nature Genet. 18, 45–48 (1998).
Iiri, T. & Bourne, H. R. Gproteins propel surprise. Nature Genet. 18, 45–48 (1998).
Clapham, D. E. & Neer, E. J. Gprotein γβ subunits. Annu. Rev. Pharmacol. Toxicol. 37, 167–203 (1997).
Berstein, G. et al. Reconstitution of agonist-stimulated phosphatidylinositol 4,5-bisphosphate hydrolysis using purified m1 muscarinic receptor, Gq/11, and phospholipase C-β1. J. Biol. Chem. 267, 8081–8088 (1992).
Farfel, Z. et al. Pseudohypoparathyroidism, a novel mutation in the βγ-contact region of Gsαimpairs receptor stimulation. J. Biol. Chem. 271, 19653–19655 (1996).
Iiri, T., Farfel, Z. & Bourne, H. R. Conditional activation defect of a human Gsαmutant. Proc. Natl Acad. Sci. USA 94, 5656–5661 (1997).
Stratton, H. F., Zhou, J., Reed, S. I. & Stone, D. E. The mating-specific Gαprotein of Saccharomyces cerevisiae downregulates the mating signal by a mechanism that is dependent on pheromone and independent of Gβγsequestration. Mol. Cell. Biol. 16, 6325–6337 (1996).
Higashijima, T., Ferguson, K. M., Sternweis, P. C., Smigel, M. D. & Gilman, A. G. Effects of Mg2+ an the βγ-subunit on the interactions of guanine nucleotides with G proteins. J. Biol. Chem. 262, 762–766 (1987).
Sternweis, P. C. The active role of beta gamma in signal transduction. Curr. Opin. Cell Biol. 6, 198–203 (1994).
Acknowledgements
This work was supported in part by a Julius Comroe Fellowship (T.I.), grants from the United States–Israel Binational Foundation and the Israeli Ministry of Health (Z.F.), and by NIH grants (H.R.B.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iiri, T., Farfel, Z. & Bourne, H. G-protein diseases furnish a model for the turn-on switch. Nature 394, 35–38 (1998). https://doi.org/10.1038/27831
Published:
Issue Date:
DOI: https://doi.org/10.1038/27831
This article is cited by
-
Molecular basis of a novel oncogenic mutation in GNAO1
Oncogene (2011)
-
Heterotrimeric G protein activation by G-protein-coupled receptors
Nature Reviews Molecular Cell Biology (2008)
-
How GPCRs hit the switch
Nature Structural & Molecular Biology (2006)
-
Mechanism of the receptor-catalyzed activation of heterotrimeric G proteins
Nature Structural & Molecular Biology (2006)
-
Probing the activation-promoted structural rearrangements in preassembled receptor–G protein complexes
Nature Structural & Molecular Biology (2006)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.