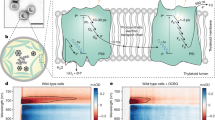
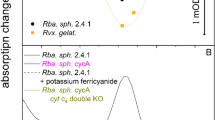
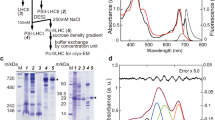
Abstract
THE primary light-induced electron transfer events of both green plant and bacterial photosynthesis result in very rapid charge separation which subsequently generates a chemical potential within the organism. There is much information concerning the primary events of bacterial photosynthesis but for green plants the details of these processes are just beginning to become available. Strong evidence based mainly on electron spin resonance and electron-nuclear double resonance studies of oxidised bacterial reaction centres favours a dimeric bacteriochlorophyll a (BChl a) structure for the primary photochemical electron donor1. The dimeric BChl a species absorbs light at 865nm in reaction centres from Rhodopseudomonas sphaeroides yielding an excited state which rapidly transfers an electron to a molecule of bacteriopheophytin a (BPh a) in <10 ps (refs 2–4). Within about 150 ps the electron is transferred to a quinone molecule which in turn transfers an electron to a secondary quinone within a few microseconds5,6. In reaction centres for which the quinones have been either extracted or chemically reduced before light excitation, BPh− back transfers an electron to (BChl a)2+ in 10–20 ns (ref. 7). Thus, the in vivo geometry of the reaction centre is such that the reverse electron transfer is 5–10 × 103 times slower than the forward reaction. We now report the first in vitro model that duplicates both the rapid light-induced charge transfer and the slow back reaction of the reaction centre.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Norris, J. R., Scheer, H. & Katz, J. J. Ann. N. Y. Acad. sci. 244, 261–280 (1975).
Kaufmann, K. J., Dutton, P. L., Netzel, T. L., Leigh, J. S. & Rentzepis, P. M. Science 188, 1301–1304 (1975).
Dutton, P. L., Kaufmann, K. J., Chance, B. & Rentzepis, P. M. FEBS Lett. 60, 275–280 (1975).
Rockley, M. G., Windsor, M. W., Cogdell, R. J. & Parson, W. W. Proc. natn. Acad. sci. U.S.A. 72, 2251–2255 (1975).
Wraight, C. A. Biochim. biophys. Acta 459, 525–531 (1977).
Vermeglio, A. & Clayton, R. K. Biochim. biophys. Acta 461, 159–165 (1977).
Parson, W. W., Clayton, R. K. & Cogdell, R. J. Biochim. biophys. Acta 387, 265–278 (1975).
Wasielewski, M. R., Smith, U. H., Cope, B. T. & Katz, J. J. J. Am. chem. Soc. 99, 4172–4173 (1977).
Wasielewski, M. R., Studier, M. H. & Katz, J. J. Proc. natn. Acad. sci. U.S.A. 73, 4282–4286 (1976).
Boxer, S. G. & Closs, G. L. J. Am. chem. Soc. 98, 5406–5408 (1976).
Seely, G. R. in The Chlorophylls (eds Vernon, L. P. & Seely, G. R.) 523–568 (Academic, New York, 1966).
Busch, G. E. & Rentzepis, P. M. Science 194, 276–283 (1976).
Borg, D. C., Fajer, J., Felton, R. H. & Dolphin, D. Proc. natn. Acad. sci. U.S.A. 67, 813–820 (1970).
Hiyama, T. & Ke, B. Biochim. biophys. Acta 267, 160–171 (1972).
Fujita, I., Davis, M. S. & Fajer, J. J. Am. chem. Soc. 100, 6280–6282 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PELLIN, M., KAUFMANN, K. & WASIELEWSKI, M. In vitro duplication of the primary light-induced charge separation in purple photosynthetic bacteria. Nature 278, 54–55 (1979). https://doi.org/10.1038/278054a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/278054a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.