Abstract
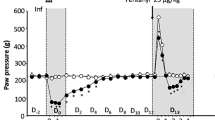
ENDOGENOUS opiate-like substances (endorphins) have been isolated from the brain in a variety of species, including man1,2. Furthermore, there is recent evidence that the brain possesses an analgesia-producing system. The distribution of endorphins is consistent and overlaps significantly with central nervous system sites such as the midbrain periaqueductal grey which produce analgesia when activated by electrical stimulation or opiate microinjection3–5. This raises the possibility that the analgesia is mediated by endorphin release. At present very little is known about the factors which trigger endorphin release. One obvious possibility is that the pain suppression system is activated by noxious stimuli. In fact, it has been observed that noxious stimuli delivered at one region of the body reduce the reported intensity of a distant noxious stimulus6. Furthermore, studies of the component neurones of this analgesia-producing system are consistent with the idea that it is activated by noxious stimuli7,8. If the action of the analgesia-producing system is mediated by endogenous morphine-like compounds, then disruption of this system by naloxone, a pure opiate antogonist, would be expected, to increase the perceived intensity of a maintained noxious stimulus. Some animal studies report no effect of naloxone alone on baseline pain thresholds9,10; in other studies, however, a shortening of latencies on hot-plate and tail-flick tests has been observed11,12. In humans, pain thresholds are unaffected by naloxone alone13,14. However, reversal of analgesia produced by electrical stimulation15,16 or by acupuncture17 has been reported. In 1965 Lasagna commented on the “curious hyper-algesic effect” of naloxone in patients with postoperative pain18. These observations suggested that clinical pain might provide a more appropriate model to study the action of endorphins. We demonstrate here that following the extraction of impacted wisdom teeth, naloxone causes a significantly greater increase in reported pain intensity than placebo, an observation consistent with the suggested participation of endorphins in an intrinsic pain suppression system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Hughes, J. et al. Nature 258, 577–579 (1975).
Simantov, R., Goodman, R., Aposhian, D. & Snyder, S. H. Brain Res. 111, 204–211 (1976).
Hiller, J. M., Pearson, J. & Simon, E. J. Res. Commun. chem. Pathol. Pharmac. 6, 1052–62 (1973).
Mayer, D. J. & Price, D. D. Pain 2, 379–404 (1976).
Lewis, V. A. & Gebhart, G. F. Brain Res. 124, 283–303 (1977).
Hardy, J. D., Wolff, H. G. & Goodell, H. J. clin. Invest. 23, 503 (1940).
Anderson, S. D., Basbaum, A. I. & Fields, H. L. Brain Res. 123, 363–368 (1977).
Lobatz, M. A., Proudfit, H. K. & Anderson, E. G. Neurosci. Abstr. 3, 296 (1977).
Goldstein, A., Pryor, G. T., Otis, L. S. & Larsen, F. Life Sci. 18, 599–604 (1976).
Berkowitz, B. A., Ngai, S. H. & Finck, A. D. Science 194, 967–968 (1976).
Jacob, J. J., Tremblay, E. C., Colombel, M. Psychopharmacologia 37, 217 (1974).
Berntson, G. G. & Walker, J. M. Brain Res. Bull. 2, 157–159 (1977).
El-Sobky, A., Dostrovsky, J. O. & Wall, P. D. Nature 263, 783–784 (1976).
Grevert, P. & Goldstein, G. Proc. natn. Acad. Sci. U.S.A. 74, 1291–94 (1977).
Adams, J. E. Pain 2, 161–166 (1976).
Hosobuchi, Y., Adams, J. E. & Linchitz, R. Science 197, 183–186 (1977).
Mayer, D. J., Price, D. D. & Rafii, A. Brain Res. 121, 360–373 (1977).
Lasagna, L. Proc. R. Soc. Med. 58, 978–983 (1965).
Scott, J. & Huskisson, E. C. Pain 2, 175–184 (1976).
Joyce, C. R. B., Zutshi, D. W., Hrubes, V. & Mason, R. M. Eur. J. clin. Pharmac. 8, 415–420 (1975).
Berkowitz, B., Finck, A. D. & Ngai, S. H. Neurosci. Abstr. 3, 286 (1977).
Goodman, L. S. & Gilman, A. The Pharmacological Basis of Therapeutics, 5th edn (Macmillan, New York, 1975).
Hayes, R. L., Harris, R. D., Wolskee, P. J. & Dubner, R. Neurosci. Abstr. 3, 483 (1977).
Oliveras, J. L., Hosobuchi, Y., Redjemi, F., Guilband, G. & Besson, J. H. Brain Res. 120, 221–229 (1977).
McNair, D. M., Lorr, M. & Droppleman, L. F. Profile of Mood States (Educational and Industrial Testing Service, San Diego, 1971).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
LEVINE, J., GORDON, N., JONES, R. et al. The narcotic antagonist naloxone enhances clinical pain. Nature 272, 826–827 (1978). https://doi.org/10.1038/272826a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/272826a0
This article is cited by
-
The neurochemical substrates of habitual and goal-directed control
Translational Psychiatry (2020)
-
Effect of a high-dose target-controlled naloxone infusion on pain and hyperalgesia in patients following groin hernia repair: study protocol for a randomized controlled trial
Trials (2015)
-
Inhibiting the breakdown of endogenous opioids and cannabinoids to alleviate pain
Nature Reviews Drug Discovery (2012)
-
Musculoskeletal events associated with the management of endocrine-responsive breast cancer
Oncology Reviews (2010)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.