Abstract
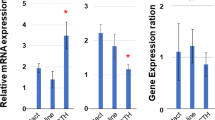
SOMATIC cell hybrids, formed by fusion of hormone-responsive and insensitive cells, have been used to evaluate the structure and genetic regulation of the hormone-sensitive adenylate cyclase system1–6. In the cell hybrids studied so far, catecholamine-sensitive adenylate cyclase activity was lost when catecholamine-responsive and insensitive cells were fused. Prostaglandin E1 (PGE1)-sensitive adenylate cyclase activity, on the other hand, was retained in hybrids between PGE1-sensitive and insensitive cells. Where examined, the presence or the absence of hormone sensitivity in the hybrids closely correlated with hormone receptor activity as measured by ligand-binding assays1–3. These observations suggest that genetic mechanisms may control the expression of hormone responsiveness at the level of the hormone receptor. To examine these patterns of regulation further, we have formed cell hybrids between rat glial tumour cells with adenylate cyclase activity responsive to β-adrenoceptor agonists7 and mouse adrenocortical tumour cells with adrenocorticotrophic hormone (ACTH)-sensitive adenylate cyclase activity8. Besides having distinct functional hormone receptors, the adenylate cyclases of parental cells exhibit marked quantitative differences in response to fluoride ion7,8. We find that the glial–adrenal hybrids retain the adrenaline-sensitive adenylate cyclase activity characteristic of the glial parent, and lose ACTH sensitivity. The level of the fluoride response, however, is characteristic of the adrenal parent. These data indicate that hormone receptor and catalytic unit activities in the adenylate cyclase system are independently regulated.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gilman, A. G. & Minna, J. D. J. biol Chem. 248, 6610–6617 (1973).
Minna, J. D. & Gilman, A. G. J. biol. Chem. 248, 6618–6625 (1973).
Brunton, L. L., Maguire, M. E., Anderson, H. J. & Gilman, A. G. J. biol. Chem. 252, 1293–1302(1977).
Hamprecht, B. & Schultz, J., Hoppe-Seyler's Z. Physiol. Chem. 354, 1633–1641 (1973).
Ayad, S. R. & Foster, S. J. Cell 3, 135–140 (1974).
Sharma, S. K., Nirenberg, M. & Klee, W. A. Proc. natn. Acad. Sci. U.S.A. 72, 590–594 (1975).
Schimmer, B. P. Biochem biophys. Acta. 252, 567–573 (1971).
Schimmer, B. P. J. biol. Chem. 247, 3134–3138 (1972).
Taunton, O. D., Roth, J. & Pastan, I. J. biol. Chem. 244, 247–253 (1969).
Benda, P., Lightbody, J., Sato, G., Levine, L. & Sweet, W. Science 161, 370–371 (1968).
Yasumura, Y., Buonassisi, V. & Sato, G. H. Cancer Res. 26, 529–535 (1966).
Schimmer, B. P. J. Cell Physiol. 74, 115–122 (1969).
Schimmer, B. P., Stevenson, L. F., ter Hofstede, C., Cheung, N. H., & Marks, A. Expl Cell Res. 86, 425–428 (1974).
Harris, H. & Cook, P. R. J. Cell Sci. 5, 121–134 (1969).
Littlefield, J. W. Proc. natn. Acad. Sci. U.S.A. 50, 568–576 (1963).
Watkins, J. in Methods in Virology 5 (eds Maramorosch, K. & Koprowski, H.) 1–32 (Academic, New York, 1971).
Littlefield, J. W. Science 145, 709–710 (1964).
Rothfels, K. H. & Siminovitch, L. Stain Tech. 33, 73–77 (1958).
Schimmer, B. P. Nature 259, 482–483 (1976).
Orly, J. & Schramm, M. Proc. natn. Acad. Sci. U.S.A. 73, 4410–4414 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SCHIMMER, B., TSAO, J. & CHEUNG, N. Regulation of adenylate cyclase activity in glial—adrenal hybrid cells. Nature 269, 162–163 (1977). https://doi.org/10.1038/269162a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/269162a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.