Abstract
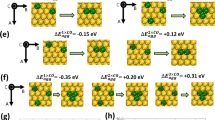
THERE is presently an extensive research effort in the field of catalysis concerned with alloy systems. Many researchers are preparing and characterising unsupported systems in order to study the metal phase in the absence of the support. A classical approach to doing this is that of simultaneous chemical reduction of the salts of a given alloy system in aqueous solution. Such reducing agents as hydrazine or sodium borohydride are frequently used. During the course of our work we found that the reduction of ruthenium salts with borohydride solutions gave a ‘metal powder’ which, when dried, violently exploded on touching it with water and sometimes by simple stimulation with a spatula. Under certain conditions a very unstable and poorly characterised ‘ruthenium hydride’ is apparently formed. A safer alternative technique is to use aqueous solutions of hydrazine for the chemical reduction of ruthenium-containing solutions.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
CUSUMANO, J. Safety in the preparation of multimetallic catalysts. Nature 247, 456 (1974). https://doi.org/10.1038/247456b0
Received:
Issue Date:
DOI: https://doi.org/10.1038/247456b0
This article is cited by
-
The early history of catalysis by gold
Gold Bulletin (2008)
-
A review on fulminating gold (Knallgold)
Gold Bulletin (2008)
-
Quantification of the abrasive wear of a gold wedding ring
Gold Bulletin (2008)
-
Fulminating gold
Gold Bulletin (2003)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.