Abstract
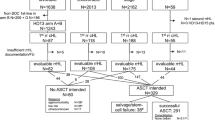
To evaluate the outcome of a large series of patients who received high-dose treatment (HDT) for follicular lymphoma (FL), 693 patients undergoing HDT (total-body irradiation (TBI)-containing regimen: 58%; autologous bone marrow (BM)/peripheral blood progenitor cells (PBPCs): 378/285 patients) were included in the study. A total of 375 patients (54%) developed recurrent lymphoma, 10-year progression-free survival (PFS) being 31%. On multivariate analysis, younger age (P=0.003) and HDT in first complete remission (CR1) (P<0.001) correlated with longer PFS. With a median follow-up of 10.3 years, 330 patients died. Ten-year overall survival (OS) from HDT was 52%. Shorter OS was associated on multivariate analysis with older age (P<0.001), chemoresistant disease (P<0.001), BM+PBPC as source of stem cells (P=0.007) and TBI-containing regimens (P=0.004). Thirty-nine patients developed secondary myelodysplastic syndrome/acute myeloid leukaemia (MDS/AML), in 34 cases having received TBI as the conditioning regimen. The 5-year non-relapse mortality (NRM) was 9%. On multivariate analysis, older age (P<0.001), refractory disease (P<0.001) and TBI (P=0.04) were associated with a higher NRM. This long follow-up study shows a plateau in the PFS curve, suggesting that a selected group of patients might be cured with HDT. On the downside, TBI-containing regimens are associated with a negative impact on survival.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Armitage JO, Weisenburger DD . New approach to classifying non-Hodgkin's lymphomas: clinical features of the major histologic subtypes. Non-Hodgkin's Lymphoma Classification Project. J Clin Oncol 1998; 16: 2780–2795.
Johnson PW, Rohatiner AZ, Whelan JS, Price CG, Love S, Lim J et al. Patterns of survival in patients with recurrent follicular lymphoma: a 20-year study from a single center. J Clin Oncol 1995; 13: 140–147.
Freedman AS, Ritz J, Neuberg D, Anderson KC, Rabinowe SN, Mauch P et al. Autologous bone marrow transplantation in 69 patients with a history of low-grade B-cell non-Hodgkin's lymphoma. Blood 1991; 77: 2524–2529.
Rohatiner AZ, Johnson PW, Price CG, Arnott SJ, Amess JA, Norton AJ et al. Myeloablative therapy with autologous bone marrow transplantation as consolidation therapy for recurrent follicular lymphoma. J Clin Oncol 1994; 12: 1177–1184.
Bastion Y, Brice P, Haioun C, Sonet A, Salles G, Marolleau JP et al. Intensive therapy with peripheral blood progenitor cell transplantation in 60 patients with poor-prognosis follicular lymphoma. Blood 1995; 86: 3257–3262.
Bierman PJ, Vose JM, Anderson JR, Bishop MR, Kessinger A, Armitage JO . High-dose therapy with autologous hematopoietic rescue for follicular low-grade non-Hodgkin's lymphoma. J Clin Oncol 1997; 15: 445–450.
Freedman AS, Neuberg D, Mauch P, Soiffer RJ, Anderson KC, Fisher DC et al. Long-term follow-up of autologous bone marrow transplantation in patients with relapsed follicular lymphoma. Blood 1999; 94: 3325–3333.
Apostolidis J, Gupta RK, Grenzelias D, Johnson PW, Pappa VI, Summers KE et al. High-dose therapy with autologous bone marrow support as consolidation of remission in follicular lymphoma: long-term clinical and molecular follow-up. J Clin Oncol 2000; 18: 527–536.
Brice P, Simon D, Bouabdallah R, Belanger C, Haioun C, Thieblemont C et al. High-dose therapy with autologous stem-cell transplantation (ASCT) after first progression prolonged survival of follicular lymphoma patients included in the prospective GELF 86 protocol. Ann Oncol 2000; 11: 1585–1590.
Gratwohl A, Baldomero H, Passweg J, Frassoni F, Niederwieser D, Schmitz N et al. Hematopoietic stem cell transplantation for hematological malignancies in Europe. Leukemia 2003; 17: 941–959.
Deconinck E, Foussard C, Milpied N, Bertrand P, Michenet P, Cornillet-LeFebvre P et al. High-dose therapy followed by autologous purged stem-cell transplantation and doxorubicin-based chemotherapy in patients with advanced follicular lymphoma: a randomized multicenter study by GOELAMS. Blood 2005; 105: 3817–3823.
Lenz G, Dreyling M, Schiegnitz E, Forstpointner R, Wandt H, Freund M et al. Myeloablative radiochemotherapy followed by autologous stem cell transplantation in first remission prolongs progression-free survival in follicular lymphoma: results of a prospective, randomized trial of the German Low-Grade Lymphoma Study Group. Blood 2004; 104: 2667–2674.
Schouten HC, Qian W, Kvaloy S, Porcellini A, Hagberg H, Johnson HE et al. High-dose therapy improves progression-free survival and survival in relapsed follicular non-Hodgkin's lymphoma: results from the randomized European CUP trial. J Clin Oncol 2003; 21: 3918–3927.
Darrington DL, Vose JM, Anderson JR, Bierman PJ, Bishop MR, Chan WC et al. Incidence and characterization of secondary myelodysplastic syndrome and acute myelogenous leukemia following high-dose chemoradiotherapy and autologous stem-cell transplantation for lymphoid malignancies. J Clin Oncol 1994; 12: 2527–2534.
Milligan DW, Ruiz De Elvira MC, Kolb HJ, Goldstone AH, Meloni G, Rohatiner AZ et al. Secondary leukaemia and myelodysplasia after autografting for lymphoma: results from the EBMT. EBMT Lymphoma and Late Effects Working Parties. European Group for Blood and Marrow Transplantation. Br J Haematol 1999; 106: 1020–1026.
Friedberg JW, Neuberg D, Stone RM, Alyea E, Jallow H, LaCasce A et al. Outcome in patients with myelodysplastic syndrome after autologous bone marrow transplantation for non-Hodgkin's lymphoma. J Clin Oncol 1999; 17: 3128–3135.
www.ebmt.org/4Registry/registry3.htmlMED-ABManual2.pdf, June 2007.
Cheson BD, Horning SJ, Coiffier B, Shipp MA, Fisher RI, Connors JM et al. Report of an international workshop to standardize response criteria for non-Hodgkin's lymphomas. NCI Sponsored International Working Group. J Clin Oncol 1999; 17: 1244–1253.
Klein JP, Rizzo JD, Zhang MJ, Keiding N . Statistical methods for the analysis and presentation of the results of bone marrow transplants. Part I: unadjusted analysis. Bone Marrow Transplant 2001; 28: 909–915.
Klein JP, Rizzo JD, Zhang MJ, Keiding N . Statistical methods for the analysis and presentation of the results of bone marrow transplants. Part 2: regression modeling. Bone Marrow Transplant 2001; 28: 1001–1011.
Colombat P, Cornillet P, Deconinck E, Tourani JM, Gardembas M, Delain M et al. Value of autologous stem cell transplantation with purged bone marrow as first-line therapy for follicular lymphoma with high tumor burden: a GOELAMS phase II study. Bone Marrow Transplant 2000; 26: 971–977.
Sebban C, Mounier N, Brousse N, Belanger C, Brice P, Haioun C et al. Standard chemotherapy with interferon compared with CHOP followed by high-dose therapy with autologous stem cell transplantation in untreated patients with advanced follicular lymphoma: the GELF-94 randomized study from the Groupe d’Etude des Lymphomes de l’Adulte (GELA). Blood 2006; 108: 2540–2544.
Rohatiner AZ, Davies AJ, Apostolidis J, Neuberg D, Matthews J, Nadler L et al. Myeloablative therapy with autologous bone marrow transplantation for follicular lymphoma at the time of second or subsequent remission. Long-term follow-up. J Clin Oncol 2007; 25: 2554–2559.
Lenz G, Dreyling M, Schiegnitz E, Haferlach T, Hasford J, Unterhalt M et al. Moderate increase of secondary hematologic malignancies after myeloablative radiochemotherapy and autologous stem-cell transplantation in patients with indolent lymphoma: results of a prospective randomized trial of the German Low Grade Lymphoma Study Group. J Clin Oncol 2004; 22: 4926–4933.
Micallef IN, Lillington DM, Apostolidis J, Amess JA, Neat M, Matthews J et al. Therapy-related myelodysplasia and secondary acute myelogenous leukemia after high-dose therapy with autologous hematopoietic progenitor-cell support for lymphoid malignancies. J Clin Oncol 2000; 18: 947–955.
Metayer C, Curtis RE, Vose J, Sobocinski KA, Horowitz MM, Bhatia S et al. Myelodysplastic syndrome and acute myeloid leukemia after autotransplantation for lymphoma: a multicenter case-control study. Blood 2003; 101: 2015–2023.
Hosing C, Munsell M, Yazji S, Andersson B, Couriel D, de Lima M et al. Risk of therapy-related myelodysplastic syndrome/acute leukemia following high-dose therapy and autologous bone marrow transplantation for non-Hodgkin's lymphoma. Ann Oncol 2002; 13: 450–459.
Krishnan A, Bhatia S, Slovak ML, Arber DA, Niland JC, Nademanee A et al. Predictors of therapy-related leukemia and myelodysplasia following autologous transplantation for lymphoma: an assessment of risk factors. Blood 2000; 95: 1588–1593.
van Besien K, Loberiza Jr FR, Bajorunaite R, Armitage JO, Bashey A, Burns LJ et al. Comparison of autologous and allogeneic hematopoietic stem cell transplantation for follicular lymphoma. Blood 2003; 102: 3521–3529.
Acknowledgements
We are indebted to all transplantation centres, physicians, nurses and data managers for their continued help in EBMT studies. A list of the transplant centres and investigators of the European Blood and Marrow Transplantation Group appears in the ‘Appendix’. SM was kindly supported by a grant from Olivia Walduck's family.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Dedicated to the memory of Felix Reyes.
Appendix
Appendix
Agnes Buzyn, Hôpital Necker, Paris, France [FR][160] 3; Roelof Willemze, Leiden University Hospital, Leiden, The Netherlands [NL][203] 7; Jane Apperley, Imperial College, Hammersmith Hospital, London, United Kingdom [GB][205] 5; Niels Jacobsen, Rigshospitalet, Copenhagen, Denmark [DK][206] 10; J Maertens, University Hospital Gasthuisberg, Leuven, Belgium [BE][209] 4; Norbert C Gorin, Hopital Saint Antoine, Paris, France [FR][213] 38; Stephen Mackinnon, Royal Free Hospital and School of Medicine, London, United Kingdom [GB][216] 1; Kurt Leibundgut, University Hospital Bern, Bern, Switzerland [CH][221] 1; Bernard Rio, Hotel Dieu, Paris, France [FR][222] 8; Antony H Goldstone, University College London Hospital, London, United Kingdom [GB][224] 5; John M Davies, Western General Hospital, Edinburgh, United Kingdom [GB][228] 1; Didier Blaise, Unité de transplantation et de thérapie cellulaire, Marseille, France [FR][230] 22; Roberto Foa, Univ. ‘La Sapienza’, Rome, Italy [IT][232] 6; Eric Deconinck, Hopital Jean Minjoz, Besancon, France [FR][233] 10; Augustin Ferrant, Cliniques Universitaires St Luc, Brussels, Belgium [BE][234] 13; A Schattenber, University Medical Center St Radboud, Nijmegen, The Netherlands [NL][237] 8; Leo F Verdonck, University Medical Centre, Utrecht, The Netherlands [NL][239] 3; Michele Baccarani, Bologna University, S Orsola-Malpighi, Bologna, Italy [IT][240] 5; Pierre Biron, Centre Leon Berard, Lyon, France [FR][241] 8; Arturo Iriondo, Hospital U Marqués de Valdecilla, Santander, Spain [ES][242] 15; H Van den Berg, Academisch Ziekenhuis bij de Universiteit, Amsterdam, The Netherlands [NL][247] 1; Oumedaly Reman, Centre Hospitalier Universitaire, Caen, France [FR][251] 1; Catherine Cordonnier, Hôpital Henri Mondor, Creteil, France [FR][252] 27; JL Harousseau, Hotel Dieu, Nantes, France [FR][253] 37; Shimon Slavin, Hadassah University Hospital, Jerusalem, Israel [IL][258] 19; Anna Sureda, Hospital de la Santa Creu i Sant Pau, Barcelona, Spain [ES][ 260] 5; JP Vernant, Hopital Pitie-Salpetriere, Paris, France [FR][262] 12; F Guilhot, Hopital La Miletrie, Poitiers, France [FR][264] 2; Gunnar Oberg, University Hospital, Uppsala, Sweden [SE][266] 3; Noel Milpied, CHU Bordeaux, Pessac, France [FR][267] 37; P Colombat, Hopital Bretonneau, Tours, France [FR][272] 10; Jacques-Olivier Bay, Fédération de Greffe de Moelle et de Therapie Cellulaire, Clermont-Ferrand, France [FR][273] 3; GH Jackson, Royal Victoria Infirmary, Newcastle- upon-Tyne, United Kingdom [GB][276] 1; JP Jouet, Hopital Claude Huriez, Lille, France [FR][277] 7; Carlos Solano, Hospital Clínico Universitario, Valencia, Spain [ES][282] 1; DW Milligan, Birmingham Heartlands Hospital, Birmingham, United Kingdom [GB][284] 3; Ignazio Majolino, Ospedale S Camillo-Forlanini, Rome, Italy [IT][287] 1; Keith Wilson, University of Wales, Cardiff, United Kingdom [GB][303] 2; Alberto Bosi, Ospedale di Careggi, Firenze, Italy [IT][304] 9; Giuseppe Leone, Universita Cattolica S Cuore, Rome, Italy [IT][307] 3; Urs Hess, Kantonsspital St Gallen, St Gallen, Switzerland [CH][324] 1; Rainer Haas, Heinrich Heine Universität, Düsseldorf, Germany [DE][390] 1; Rosanna Scimè, Ospedale V Cervello, Palermo, Italy [IT][392] 4; Filomena Floristan, Hospital de Cruces, Barakaldo (Bilbao), Spain [ES][393] 1; Tapio Nousiainen, Kuopio University Hospital, Kuopio, Finland [FI][396] 1; Tapani Ruutu, Helsinki University Central Hospital, Helsinki, Finland [FI][515] 2; Martin Hildebrandt, Charité Universitätsmedizin Berlin, Berlin, Germany [DE][518] 1; Nicole Gratecos, Hôpital de l’ARCHET I, Nice, France [FR][523] 1; M Janvier, Centre Rene Huguenin, Saint Cloud, France [FR][551] 3; Harry Schouten, University Hospital Maastricht, Maastricht, The Netherlands [NL][ 565] 8; Bjarne Jensen, Herlev Hospital Anker, Herlev, Denmark [DK][568] 3; GJ Ossenkoppel, VU University Medical Centere, Amsterdam, The Netherlands [NL][588] 1; Axel A Fauser, Klinik für Knochenmarktransplantation, Idar -Oberstein, Germany [DE][592] 1; W Feremans, ULB—Hopital Erasme, Brussels, Belgium [BE][596] 2; Fabio Benedetti, Policlinico GB Rossi, Verona, Italy [IT][623] 3; R Schots, University Hospital VUB, Brussels, Belgium [BE][630] 1; Elli Koivunen, Tampere University Hospital, Tampere, Finland [FI][635] 1; Norbert Ifrah, CHRU, Angers, France [FR][650] 6; Tiziano Barbui, Ospedale Bergamo, Bergamo, Italy [IT][658] 2; Thierry de Revel, Hôpital Percy, Clamart, France [FR][665] 3; Jean HenriBourhis, Institut Gustave Roussy, Villejuif, France [FR][666] 3; Mauricette Michallet, Hopital E Herriot, Lyon, France [FR][671] 9; NH Russell, Nottingham City Hospital, Nottingham, United Kingdom [GB][717] 2; Vladimir Koza, Charles University Hospital, Pilsen, Czech Republic [CZ][718] 3; J Besalduch, Hospital Universitari Son Dureta, Palma de Mallorca, Spain [ES][722] 1; Dolores Caballero, Hospital Clínico, Salamanca, Spain [ES][727] 2; M Komarnicki, K Marcinkowski University of Medical Science, Poznan, Poland [PL][730] 1; Anders Wahlin, Umea University Hospital, Umea, Sweden [SE][731] 2; José M Moraleda Jimenez ,Hospital Morales Meseguer, Murcia, Spain [ES][735] 6; Elisabeth Koller, Hanuschkrankenhaus, Vienna, Austria [AT][743] 2; Marek Trneny, Charles University Hospital, Prague, Czech Republic [CZ][745] 3; Jean-Pierre Lotz, Hôpital Tenon, Paris, France[FR][747] 1; GJ Mufti, GKT School of Medicine, London, United Kingdom [GB][763] 1; John Gribben, St Bartholomew’s and The Royal London Hospital, London, United Kingdom [GB][768] 114; Joerg Schubert, University of Saarland, Homburg, Germany [DE][785] 1; Massimo F Martelli, University of Perugia, Perugia, Italy [IT][794] 21; Francesco Rodeghiero, S Bortolo Hospital, Vicenza, Italy [IT][797] 2; C Gisselbrecht, Hôpital St Louis, Paris, France [FR][805] 49; Jean Francois Rossi, CHU Lapeyronie, Montpellier, France [FR][926] 7; H Tilly, Centre Henri Becqueral, Rouen, France [FR][941] 11; Ghandi Damaj, University of Amiens: CHU Amiens, Amiens, France [FR][955] 1; Alain Delmer, Hôpital Robert Debre, Reims, France [FR][959] 6; Laurent Degos, Institut Universitaire d’Hématologie, Paris, France [FR][960] 2; Dominique Bordessoule, CHRU Limoges, Limoges, France [FR][977] 2.
Rights and permissions
About this article
Cite this article
Montoto, S., Canals, C., Rohatiner, A. et al. Long-term follow-up of high-dose treatment with autologous haematopoietic progenitor cell support in 693 patients with follicular lymphoma: an EBMT registry study. Leukemia 21, 2324–2331 (2007). https://doi.org/10.1038/sj.leu.2404850
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404850
Keywords
This article is cited by
-
Optimized BEAC conditioning regimen improves clinical outcomes of autologous hematopoietic stem cell transplantation in non-Hodgkin lymphomas
International Journal of Hematology (2024)
-
Durable benefit of rituximab maintenance post-autograft in patients with relapsed follicular lymphoma: 12-year follow-up of the EBMT lymphoma working party Lym1 trial
Bone Marrow Transplantation (2021)
-
Outcome of allogeneic hematopoietic stem cell transplantation for follicular lymphoma relapsing after autologous transplantation: analysis of the Japan Society for Hematopoietic Cell Transplantation
Bone Marrow Transplantation (2021)
-
Strategies to improve outcomes of autologous hematopoietic cell transplant in lymphoma
Bone Marrow Transplantation (2019)
-
The role of stem cell transplantation in the management of relapsed follicular lymphoma in the era of targeted therapies
Bone Marrow Transplantation (2019)