Abstract
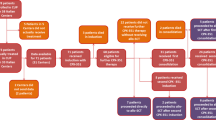
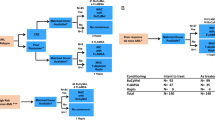
Between 1988 and 2002, 758 children with acute myeloid leukaemia (AML) were treated on Medical Research Council (MRC) AML 10 and AML 12. MRC AML 10 tested the role of bone marrow transplantation following four blocks of intensive chemotherapy and found that while both allogeneic bone marrow transplant (allo-BMT) and autologous bone marrow transplant (A-BMT) significantly reduced the relapse risk (RR), this did not translate into a significant improvement in overall survival (OS). A risk group stratification based on cytogenetics and response to the first course of chemotherapy derived from MRC AML 10 was used to deliver risk-directed therapy in MRC AML 12. Allo-BMT was limited to standard and poor risk patients and A-BMT was not employed. Instead, the benefit of an additional block of treatment was tested by randomising children to receive either four or five blocks of treatment in total. While the results of MRC AML 12 remain immature, there appears to be no survival advantage for a fifth course of treatment. The 5 year OS, disease-free survival (DFS), event-free survival (EFS) and RR in MRC AML 12 are 66, 61, 56 and 35%, respectively; at present superior to MRC AML 10, which had a 5-year OS, DFS, EFS and RR of 58, 53, 49 and 42%, respectively. MRC AML trials employ a short course of triple intrathecal chemotherapy alone for CNS-directed treatment and CNS relapse is uncommon. Improvements in supportive care have contributed to improved outcomes and the number of deaths in remission fell between trials. Anthracycline-related cardiotoxicity remains a concern and the current MRC AML 15 trial tests the feasibility of reducing anthracycline dosage without compromising outcome by comparing standard MRC anthracycline-based consolidation with high-dose ara-C. MRC studies suggest that the role of allo-BMT is limited in 1st CR and that there may be a ceiling of benefit from current or conventional chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rees JKH, Gray RG, Swirsky D, Hayhoe FGJ, for the Medical Research Council's Working Parties for Therapeutic Trials in Adult and Childhood Leukaemia. Principal results of the Medical Research Council's 8th Acute Myeloid Leukaemia Trial. Lancet 1986; ii: 1236–1241.
Marcus RE, Catovsky D, Prentice HG, Newland AC, Chessells JM, Stevens RM et al. Intensive Induction and Consolidation Chemotherapy for Adults and Children with Acute Myeloid Leukaemia (AML) Joint AML Trial 1982–1985. In: Büchner Th, Schellong G, Hiddemann W, Urbanitz D, Ritter J, (eds). Acute Leukaemias. Prognostic Factors and Treatment Strategies, Haematology and Blood Transfusion, Vol. 30. Berlin-Heidelberg-New York, Tokyo: Springer-Verlag, 1987.
Rees JKH, Gray RG, Wheatley K, on behalf of the MRC Leukaemia in Adults Working Party. Dose intensification in acute myeloid leukaemia: greater effectiveness at lower cost. Principal report of the Medical Research Council's AML9 study. Br J Haematol 1996; 94: 89–98.
Worsley AM, Catovsky D, Goldman JM, Galton DAG . New combination chemotherapy for relapsed acute myeloid leukaemia. Lancet 1984; I: 1232.
Preisler H . High-dose Ara-C as the initial treatment of poor risk patients with acute non-lymphocytic leukaemia. A leukaemia intergroup study. J Am Soc Clin Oncol 1987; 5: 75–82.
Herzig RH, Lazarus HM, Wolff SN, Phillips GL, Herzig GP . High-dose Ara-C therapy with and without anthracycline antibiotics for remission re-induction of acute non-lymphoblastic leukaemia. J Clin Oncol 1985; 3: 992–997.
Brito-Babapulle F, Catovsky D, Slocombe G, Newland AC, Marcus RE, Goldman JM et al. Phase II study of mitoxantrone and cytarabine in acute myeloid leukaemia. Cancer Treat Rep 1987; 71: 161–163.
Hiddemann W, Kreutzmann H, Straif K, Ludwig WD, Mertelsmann R, Donhuijsen-Ant R et al. High dose cytosine arabinoside and mitoxantrone: a highly effective regimen in refractory acute myeloid leukaemia. Blood 1987; 69: 744–749.
Peters WG, Willemze R, Colly LP . Results of induction and consolidation treatment with intermediate and high-dose ara-C and m-AMSA-containing regimens in patients with primarily failed or relapsed acute leukaemia and non-Hodgkin's lymphoma. Scand J Haem 1986; 36: 7–16.
Willemze R, Zwaan FE, Colpri G, Kenning JJ . High-dose cytosine arabinoside in the management of refractory acute leukaemia. Scand J Haem 1982; 29: 141–146.
Arlin Z, Case DC, Moore J, Wiernik P, Feldman E, Saletan S et al. Randomized multi-centre trial of cytosine arabinoside with mitoxantrone or daunorubicin in previously untreated adult patients with acute non-lymphoblastic leukaemia (ANLL). Leukaemia 1990; 4: 177–183.
Kaspers GJL, Creutzig U . Pediatric acute myeloid leukaemia: International progress and future directions. Leukaemia 2005; 19: 2025–2029.
Wheatley K, Burnett AK, Goldstone AH, Gray RG, Hann IM, Harrison CJ, et al, on behalf of the United Kingdom Medical Research Council's Adult and Childhood Leukaemia Working Parties. A simple, robust, validated and highly predictive index for the determination of risk-directed therapy in acute myeloid leukaemia derived from the MRC AML 10 trial. Br J Haematol 1999; 107: 69–79.
Riley LC, Hann IM, Wheatley K, Stevens RF, on behalf of the MRC Childhood Leukaemia Working Party. Treatment-related deaths during induction and first remission of acute myeloid leukaemia in children treated on the Tenth Medical Research Council Acute Myeloid Leukaemia Trial (MRC AML10). Br J Haematol 1999; 106: 436–444.
Woods WG, Kobrinsky N, Buckley J, Neudorf S, Sanders J, Miller L et al. Intensively timed induction therapy followed by autologous or allogeneic bone marrow transplantation for children with acute myeloid leukaemia or myelodysplastic syndrome: a Children's Cancer Group pilot study. J Clin Oncol 1993; 8: 1448–1457.
Amadori S, Testi AM, Arico M, Comelli A, Giuliano M, Madon E et al. Prospective comparative study of bone marrow transplantation and post remission chemotherapy for childhood acute myelogenous leukaemia. The Associazione Italiana Ematologia ed Oncologia Pediatrica Cooperative Group. J Clin Oncol 1993; 6: 1046–1054.
Ravindranath Y, Yeager AM, Chang MN, Steuber CP, Krischer J, Graham Pole J et al. Autologous bone marrow transplantation versus intensive consolidation chemotherapy for acute myeloid leukaemia in childhood. N Engl J Med 1996; 334: 1428–1434.
Woods WG, Neudorf S, Gold S, Sanders J, Buckley JD, Barnard DR et al. A comparison of allogeneic bone marrow transplantation, autologous bone marrow transplantation, and aggressive chemotherapy in children with acute myeloid leukaemia in remission: a report from the children's cancer group. Blood 2001; 97: 56–62.
Bleakley M, Lau L, Shaw PJ, Kaufman A . Bone marrow transplantation for paediatric AML in first remission: a systematic review and meta-analysis. Bone Marrow Transplant 2002; 10: 843–852.
Creutzig U, Reinhardt D, Zimmermann M, Klingebiel T, Gadner H . Intensive Chemotherapy vs BMT in pediatric AML – a matter of controversies. Blood 2001; 97: 3671–3672.
Wheatley K . Current controversies: which patients with acute myeloid leukaemia should receive a bone marrow transplantation? A statistician's view. Br J Haematol 2002; 118: 351–356.
Creutzig U, Ritter J, Zimmermann M, Schellong G . Does cranial irradiation reduce the risk for bone marrow relapse in acute myelogenous leukaemia. Unexpected results of the Childhood Acute Myelogenous Leukaemia Study BFM-87. J Clin Oncol 1993; 11: 279–286.
Stevens RF, Hann IM, Wheatley K, Gray RG, on behalf of the MRC Childhood Leukaemia Working Party. Marked improvements in outcome with chemotherapy alone in paediatric acute myeloid leukaemia: results of the United Kingdom Medical Research Council's 10th AML trial. Br J Haematol 1998; 101: 130–140.
Acknowledgements
The MRC/UK Childhood Leukaemia Working Party acknowledge the very significant contribution to MRC AML 10 and subsequent trials of Dr Richard Stevens (Deceased). For Acknowledgements refer to Supplementary Information.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Supplementary Information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu).
Supplementary information
Rights and permissions
About this article
Cite this article
Gibson, B., Wheatley, K., Hann, I. et al. Treatment strategy and long-term results in paediatric patients treated in consecutive UK AML trials. Leukemia 19, 2130–2138 (2005). https://doi.org/10.1038/sj.leu.2403924
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403924
Keywords
This article is cited by
-
Development and validation of a promising 5-gene prognostic model for pediatric acute myeloid leukemia
Molecular Biomedicine (2024)
-
Treatment outcomes of pediatric acute myeloid leukemia: a retrospective analysis from 1996 to 2019 in Taiwan
Scientific Reports (2021)
-
Hematopoietic stem cell transplantation for children with acute myeloid leukemia—results of the AML SCT-BFM 2007 trial
Leukemia (2020)
-
Outcome of Acute Myeloid Leukemia in Children Adolescents and Young Adults Treated with an Uniform Protocol in Casablanca, Morocco
Indian Journal of Hematology and Blood Transfusion (2019)
-
Successes and challenges in the treatment of pediatric acute myeloid leukemia: a retrospective analysis of the AML-BFM trials from 1987 to 2012
Leukemia (2018)