Abstract
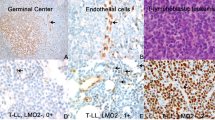
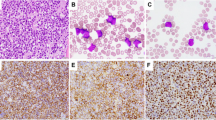
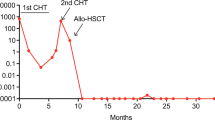
Anaplastic large cell lymphoma (ALCL) harbors the reciprocal chromosomal translocation t(2;5)(p23;q35) in approximately 80% of the cases. The genes involved are nucleophosmin (NPM) and anaplastic lymphoma kinase (ALK) and the resulting chimeric NPM–ALK protein is thought to play a key role in the pathogenesis of t(2;5) positive ALCL. Few data on bone marrow (BM) involvement in ALCL have been published and they mostly rely on morphological examination of BM smears. We studied 52 ALCL for NPM–ALK expression by RT-PCR: 47/52 biopsies were positive. In 41 of the 47 cases we obtained the BM at diagnosis and investigated the prevalence of minimal BM infiltration by RT-PCR and real-time PCR. Minimal disseminated disease was positive in 25/41 patients (61%), of whom six had morphologically infiltrated BM. Survival analysis demonstrated a 5-year progression-free survival of 41±11% for patients with molecularly positive BM vs 100% for patients with negative BM (P= 0.001). These results suggest that minimal BM involvement at diagnosis is a common event in pediatric ALCL and that minimal BM disease monitoring could identify patients at risk of relapse.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Falini B, Pileri S, Pizzolo G, Durkop H, Flenghi L, Stirpe F et al. CD30 (Ki-1) molecole: a new cytokine receptor of the tumor necrosis factor receptor superfamily as a tool for diagnosis and immunotherapy. Blood 1995; 85: 1–14.
Stansfeld AG, Diebold J, Kapanci Y, Rilke F, Kelenyi G, Sundstrom C et al. Updated Kiel classification for lymphomas. Lancet 1988; I: 292–293.
Benharroch D, Bedoyan Z, Lamant L, Amin C, Brugieres L, Terrier-Lacombe MJ et al. ALK-positive lymphoma: a single disease with a broad spectrum of morphology. Blood 1998; 91: 2076–2084.
Rimokh R, Magaud JP, Berger F, Samarut J, Coiffier B, Germain D et al. A translocation involving a specific breakpoint (q35) on chromosome 5 is characteristic of anaplastic large cell lymphoma (‘Ki-1 lymphoma’). Br J Haematol 1989; 71: 31–36.
Lamant L, Meggetto F, Saati T, Brugieres L, de Paillerets BB, Dastague N et al. High incidence of the t(2;5)(p23;q35) translocation in anaplastic large cell lymphoma and its lack of detection in Hodgkin's disease. Comparison of cytogenetic analysis, reverse transcriptase-polymerase chain reaction, and P-80 immunostaining. Blood 1996; 87: 284–291.
Morris SW, Naeve C, Mathew P, James PL, Kirstein MN, Cui X et al. ALK, the chromosome 2 gene locus altered by the t(2;5) in non-Hodgkin's lymphoma, encodes a novel neural receptor tyrosine kinase that is highly related to leukocyte tyrosine kinase (LTK). Oncogene 1997; 14: 2175–2188.
Iwahara T, Fujimoto J, Wen D, Cupples R, Bucay N, Arakawa T et al. Molecular characterization of ALK, a receptor tyrosine kinase expressed specifically in the nervous system. Oncogene 1997; 14: 439–449.
Fujimoto J, Shiota M, Iwahara T, Seki N, Satoh H, Mori S et al. Characterization of the transforming activity of p80, a hyperphosphorilated protein in a Ki-1 lymphoma cell line with chromosomal translocation t(2;5). Proc Natl Acad Sci USA 1996; 93: 4181–4186.
Kuefer MU, Look AT, Pulford K, Behm FG, Pattengale PK, Mason DY et al. Retrovirus-mediated gene transfer of NPM–ALK causes lymphoid malignancy in mice. Blood 1997; 90: 2901–2910.
Falini B, Pileri S, Zanzini PL, Carbone A, Zagonel V, Wolf-Peeters C et al. ALK+ lymphoma: clinico-pathological findings and outcome. Blood 1999; 93: 2697–2706.
Fraga M, Brousset P, Schlaifer D, Payen C, Robert A, Rubie H et al. Bone marrow involvement in anaplastic large cell lymphoma. Am J Clin Pathol 1995; 103: 82–89.
Seidemann K, Tiemann M, Schrappe M, Yakisan E, Simonitsch I, Janka-Schaub G et al. Short-pulse B-non-Hodgkin lymphoma-type chemotherapy is efficacious treatment for pediatric anaplastic large cell lymphoma: a report of the Berlin–Frankfurt–Munster Group Trial NHL-BFM 90. Blood 2001; 97: 3699–3706.
Murphy SB . Classification, staging and end results of treatment of childhood non-Hodgkin's lymphomas: dissimilarities from lymphomas in adults. Semin Oncol 1980; 7: 332–339.
Brugières L, Le Deley MC, Pacquement H, Meuerian-Bedoyan Z, Terrier-Lacombe MJ, Robert A et al. CD 30+ anaplastic large-cell lymphoma in children: analysis of 82 patients enrolled in two consecutive studies of the French Society of Pediatric Oncology. Blood 1998; 92: 3591–3598.
Kaplan EL, Meier P . Non parametric estimation from incomplete observation. J Am Stat Assoc 1958; 53: 457–481.
Peto R, Peto J . Asymptotically efficient rank invariant test procedures. J R Stat Soc A 1972; 135: 185–198.
Cox DR . Regression models and life tables. J R Stat Soc 1972; 34: 187–220.
Stein H, Dallenbach F . Diffuse large cell lymphomas of B and T cell type. In: Knowles DM (ed). Neoplastic Hematopathology. Williams and Wilkins: Baltimore, 1992, pp 675–714.
Gerard CJ, Olsson K, Ramanathan R, Reading C, Hanania EG . Improved quantitation of minimal residual disease in multiple myeloma using real-time polymerase chain reaction and plasmid-DNA complementary determining region III standards. Cancer Res 1998; 58: 3957–3964.
Marcucci G, Livak KJ, Bi W, Strout MP, Bloomfield CD, Caligiuri MA et al. Detection of minimal residual disease in patients with AML1/ETO-associated acute myeloid leukemia using a novel quantitative reverse transcription polymerase chain reaction assay. Leukemia 1998; 12: 1482–1489.
Mensink E, Van de Locht A, Schattenberg A, Linders E, Schaap N, Geurts van Kessel A et al. Quantitation of minimal residual disease in Philadelphia chromosome positive chronic myeloid leukaemia using real-time quantitative RT-PCR. Br J of Haematol 1998; 102: 768–774.
Viehmann S, Teigler-Schlegel A, Bruch J, Langebrake C, Reinhardt D, Harbott J . Monitoring of minimal residual disease (MRD) by real-time quantitative reverse transcription PCR (RQ-RT-PCR) in childhood acute myeloid leukaemia with AML1/ETO rearrangement. Leukemia 2003; 17: 1130–1136.
Acknowledgements
This work was supported by Fondazione Città della Speranza, by a grant from MIUR-CNR and by AIL (Associazione Italiana contro le Leucemie). L Mussolin is a fellow of Fondazione Città della Speranza. We thank Gloria Tridello for the statistical analysis, Ilaria Zecchini for data collection and management and Angelica Zin for technical support. We are grateful to the clinicians of the Italian Association of Pediatric Hematology and Oncology (AIEOP) for providing biological samples and patient information.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu/).
Supplementary information
Rights and permissions
About this article
Cite this article
Mussolin, L., Pillon, M., d'Amore, E. et al. Prevalence and clinical implications of bone marrow involvement in pediatric anaplastic large cell lymphoma. Leukemia 19, 1643–1647 (2005). https://doi.org/10.1038/sj.leu.2403888
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403888
Keywords
This article is cited by
-
Prognostic impact of minimal disseminated disease and immune response to NPM-ALK in Japanese children with ALK-positive anaplastic large cell lymphoma
International Journal of Hematology (2018)
-
Anaplastic large cell lymphoma-propagating cells are detectable by side population analysis and possess an expression profile reflective of a primitive origin
Oncogene (2015)