Abstract
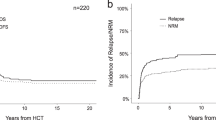
This report describes the long-term follow-up data of three consecutive Dutch Childhood Oncology Group acute myeloid leukemia (AML) protocols. A total of 303 children were diagnosed with AML, of whom 209 were eligible for this report. The first study was the AML-82 protocol. Results were inferior (5-year probability of overall survival (pOS) 31%) to other available regimes. Study AML-87 was based on the BFM-87 protocol, with prophylactic cranial irradiation in high-risk patients only, and without maintenance therapy. This led to a higher cumulative incidence of relapse than that reported by the Berlin–Frankfurt–Münster (BFM), but survival was similar (5-year pOS 47%), suggesting successful retrieval at relapse. The subsequent study AML-92/94 consisted of a modified BFM-93 protocol, that is, without maintenance therapy and prophylactic cranial irradiation. However, all patients were to be transplanted (auto- or allogeneic), although compliance was poor. Antileukemic efficacy was offset by an increase in the cumulative incidence of nonrelapse mortality, especially in remission patients, and survival did not improve (5-year pOS 44%). Our results demonstrate that outcome in childhood AML is still unsatisfactory, and that further intensification of therapy carries the risk of enhanced toxicity. Our patients are currently included in the MRC AML studies, based on the results of their AML 10 trial.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Van Der Does-Van Den Berg A, Van Wering ER, Van Zanen GE . Acute niet-lymfatische leukemieën bij kinderen in Nederland (1972–1977). Tijdschr Kindergeneeskd 1978; 46: 146–153.
Kamps WA, Veerman AJ, Van Wering ER, Van Weerden JF, Slater R, Does-van den Berg A . Long-term follow-up of Dutch Childhood Leukemia Study Group (DCLSG) protocols for children with acute lymphoblastic leukemia, 1984–1991. Leukemia 2000; 14: 2240–2246.
Van Der Does-Van Den Berg A, Hählen K, Colly LP, Vossen JM . Treatment of childhood acute nonlymphocytic leukemia with high-dose cytosine-arabinoside, 6-thioguanine and doxorubicin without maintenance therapy: pilot study ANLL-80 of the Dutch Childhood Leukemia Study Group. Pediatr Hematol Oncol 1988; 5: 93–102.
Slats AM, Egeler RM, Van Der Does-Van Den Berg A, Korbijn C, Hählen K, Kamps WA et al. Causes of death – other than progressive leukemia – in childhood acute lymphoblastic (ALL) and myeloid leukemia (AML): the Dutch Childhood Oncology Group experience. Leukemia 2005; 19: 537–544.
Weinstein HJ, Mayer RJ, Rosenthal DS, Camitta BM, Coral FS, Nathan DG et al. Treatment of acute myelogenous leukemia in children and adults. N Engl J Med 1980; 303: 473–478.
Creutzig U, Ritter J, Schellong G, for the AML-BFM Study Group. Identification of two risk groups in childhood acute myelogenous leukemia after therapy intensification in study AML-BFM-83 as compared with study AML-BFM-78. Blood 1990; 75: 1932–1940.
Creutzig U, Ritter J, Zimmermann M, Schellong G . Does cranial irradiation reduce the risk for bone marrow relapse in acute myelogenous leukemia? Unexpected results of the Childhood Acute Myelogenous Leukemia Study BFM-87. J Clin Oncol 1993; 11: 279–286.
Hählen K, Van Der Does-Van Den Berg A, Colly LP, Smets LA, Taminiau JA, Vossen JM . Treatment of childhood acute nonlymphocytic leukemia with individually scheduled high doses of cytarabine: preliminary results of study ANLL-82 of the Dutch Childhood Leukemia Study Group (DCLSG). Haematol Blood Transfus 1987; 30: 389–392.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Proposed revised criteria for the classification of acute myeloid leukemia. A report of the French–American–British Cooperative Group. Ann Intern Med 1985; 103: 620–625.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Criteria for the diagnosis of acute leukemia of megakaryocyte lineage (M7). A report of the French–American–British Cooperative Group. Ann Intern Med 1985; 103: 460–462.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Proposal for the recognition of minimally differentiated acute myeloid leukaemia (AML-MO). Br J Haematol 1991; 78: 325–329.
Creutzig U, Zimmermann M, Ritter J, Reinhardt D, Hermann J, Henze G, et al., for the AML-BFM Study Group. Treatment strategy and long-term results in pediatric patients treated in four consecutive AML-BFM trials. Leukemia, this issue.
Weinstein HJ, Mayer RJ, Rosenthal DS, Coral FS, Camitta BM, Gelber RD . Chemotherapy for acute myelogenous leukemia in children and adults: VAPA update. Blood 1983; 62: 315–319.
Inati A, Sallan SE, Cassady JR, Hitchcock-Bryan S, Clavell LA, Belli JA et al. Efficacy and morbidity of central nervous system ‘prophylaxis’ in childhood acute lymphoblastic leukemia: eight years’ experience with cranial irradiation and intrathecal methotrexate. Blood 1983; 61: 297–303.
Reinhardt D, Thiele C, Creutzig U . Neuropsychological sequelae in children with AML treated with or without prophylactic CNS-irradiation. Klin Pädiatr 2002; 214: 22–29.
Leung W, Hudson MM, Strickland DK, Phipps S, Srivastava DK, Ribeiro RC et al. Late effects of treatment in survivors of childhood acute myeloid leukemia. J Clin Oncol 2000; 18: 3273–3279.
Champlin R, Jacobs A, Gale RP, Boccia R, Elashoff R, Foon K et al. Prolonged survival in acute myelogenous leukaemia without maintenance chemotherapy. Lancet 1984; 1: 894–896.
Wells RJ, Woods WG, Buckley JD, Odom LF, Benjamin D, Bernstein I et al. Treatment of newly diagnosed children and adolescents with acute myeloid leukemia: a Childrens Cancer Group study. J Clin Oncol 1994; 12: 2367–2377.
Perel Y, Auvrignon A, Leblanc T, Vannier JP, Michel G, Nelken B et al. Impact of addition of maintenance therapy to intensive induction and consolidation chemotherapy for childhood acute myeloblastic leukemia: results of a prospective randomized trial, LAME 89/91. J Clin Oncol 2002; 20: 2774–2782.
Abbott BL, Rubnitz JE, Tong X, Srivastava DK, Pui CH, Ribeiro RC et al. Clinical significance of central nervous system involvement at diagnosis of pediatric acute myeloid leukemia: a single institution's experience. Leukemia 2003; 17: 2090–2096.
Creutzig U, Ritter J, Zimmermann M, Hermann J, Gadner H, Sawatzki DB et al. Idarubicin improves blast cell clearance during induction therapy in children with AML: results of study AML-BFM 93. Leukemia 2001; 15: 348–354.
Creutzig U, Ritter J, Zimmermann M, Reinhardt D, Hermann J, Berthold F et al. Improved treatment results in high-risk pediatric acute myeloid leukemia patients after intensification with high-dose cytarabine and mitoxantrone: results of Study Acute Myeloid Leukemia-Berlin–Frankfurt–Münster 93. J Clin Oncol 2001; 19: 2705–2713.
Berman E, Heller G, Santorsa J, McKenzie S, Gee T, Kempin S et al. Results of a randomized trial comparing idarubicin and cytosine arabinoside with daunorubicin and cytosine arabinoside in adult patients with newly diagnosed acute myelogenous leukemia. Blood 1991; 77: 1666–1674.
Wiernik PH, Banks PL, Case Jr DC, Arlin ZA, Periman PO, Todd MB et al. Cytarabine plus idarubicin or daunorubicin as induction and consolidation therapy for previously untreated adult patients with acute myeloid leukemia. Blood 1992; 79: 313–319.
Vogler WR, Velez-Garcia E, Weiner RS, Flaum MA, Bartolucci AA, Omura GA et al. A phase III trial comparing idarubicin and daunorubicin in combination with cytarabine in acute myelogenous leukemia: a Southeastern Cancer Study Group Study. J Clin Oncol 1992; 10: 1103–1111.
Sackmann-Muriel F, Fernandez-Barbieri MA, Santarelli MT, Matus-Ridley M, Rosso A, Negri-Aranguren P et al. Results of treatment with an intensive combination induction regimen containing idarubicin in children with acute myeloblastic leukemia: preliminary report of the Argentine Group for Treatment of Acute Leukemia. Semin Oncol 1993; 20: 34–38.
Reid JM, Pendergrass TW, Krailo MD, Hammond GD, Ames MM . Plasma pharmacokinetics and cerebrospinal fluid concentrations of idarubicin and idarubicinol in pediatric leukemia patients: a Children's Cancer Study Group report. Cancer Res 1990; 50: 6525–6528.
Casazza AM . Experimental evaluation of anthracycline analogs. Cancer Treat Rep 1979; 63: 835–844.
Hiddemann W, Kreutzmann H, Straif K, Ludwig WD, Mertelsmann R, Donhuijsen-Ant R et al. High-dose cytosine arabinoside and mitoxantrone: a highly effective regimen in refractory acute myeloid leukemia. Blood 1987; 69: 744–749.
Amadori S, Testi AM, Aricò M, Comelli A, Giuliano M, Madon E et al. Prospective comparative study of bone marrow transplantation and post-remission chemotherapy for childhood acute myelogenous leukemia. J Clin Oncol 1993; 11: 1046–1054.
Tiedemann K, Waters KD, Tauro GP, Tucker D, Ekert H . Results of intensive therapy in childhood acute myeloid leukemia, incorporating high-dose melphalan and autologous bone marrow transplantation in first complete remission. Blood 1993; 82: 3730–3738.
Lehrnbecher T, Varwig D, Kaiser J, Reinhardt D, Klingebiel T, Creutzig U . Infectious complications in pediatric acute myeloid leukemia: analysis of the prospective multi-institutional clinical trial AML-BFM 93. Leukemia 2004; 18: 72–77.
Ravindranath Y, Yeager AM, Chang MN, Steuber CP, Krischer J, Graham-Pole J, et al., for the Pediatric Oncology Group. Autologous bone marrow transplantation versus intensive consolidation chemotherapy for acute myeloid leukemia in childhood. N Engl J Med 1996; 334: 1428–1434.
Creutzig U, Reinhardt D . Current controversies: which patients with acute myeloid leukaemia should receive a bone marrow transplantation? – a European view. Br J Haematol 2002; 118: 365–377.
Chen AR, Alonzo TA, Woods WG, Arceci RJ . Current controversies: which patients with acute myeloid leukaemia should receive a bone marrow transplantation? – an American view. Br J Haematol 2002; 118: 378–384.
Stevens RF, Hann IM, Wheatley K, Gray RG, on behalf of the MRC Childhood Leukaemia Working Party. Marked improvements in outcome with chemotherapy alone in paediatric acute myeloid leukemia: results of the United Kingdom Medical Research Council's 10th AML trial. Br J Haematol 1998; 101: 130–140.
Sievers EL, Lange BJ, Alonzo TA, Gerbing RB, Bernstein ID, Smith FO et al. Immunophenotypic evidence of leukemia after induction therapy predicts relapse: results from a prospective Children's Cancer Group study of 252 acute myeloid leukemia patients. Blood 2002; 101: 3398–3406.
Feller N, Van der Pol MA, Van Stijn A, Weijers GW, Westra AH, Evertse BW et al. MRD parameters using immunophenotypic detection methods are highly reliable in predicting survival in acute myeloid leukaemia. Leukemia 2004; 18: 1380–1390.
Zwaan ChM, Meshinchi S, Radich JP, Veerman AJP, Huismans DR, Munske L et al. FLT3 internal tandem duplication in 234 children with acute myeloid leukemia (AML): prognostic significance and relation to cellular drug resistance. Blood 2003; 102: 2387–2394.
George B, Mathews V, Poonkuzhali B, Shaji RV, Srivastava A, Chandy M . Treatment of children with newly diagnosed acute promyelocytic leukemia with arsenic trioxide: a single center experience. Leukemia 2004; 18: 1587–1590.
Ravindranath Y . Down syndrome and acute myeloid leukemia: the paradox of increased risk for leukemia and heightened sensitivity to chemotherapy. J Clin Oncol 2003; 21: 3385–3387.
Zwaan ChM, Reinhardt D, Corbacioglu S, Van Wering ER, Bökkerink JP, Tissing WJ et al. Gemtuzumab ozogamicin: first clinical experiences in children with relapsed/refractory acute myeloid leukemia treated on compassionate use basis. Blood 2003; 101: 3868–3871.
Appelbaum FR . New targets for therapy in acute myeloid leukemia. Leukemia 2003; 17: 492–495.
Acknowledgements
We thank all physicians and hospitals for participating in the Dutch Childhood Oncology Study Group AML studies, as well as the technicians and staff from the DCOG central laboratory and data management office. Statistical advice and support was given by R Brand (Leiden University Medical Center, Leiden, The Netherlands).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu).
Rights and permissions
About this article
Cite this article
Kardos, G., Zwaan, C., Kaspers, G. et al. Treatment strategy and results in children treated on three Dutch Childhood Oncology Group acute myeloid leukemia trials. Leukemia 19, 2063–2071 (2005). https://doi.org/10.1038/sj.leu.2403873
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403873
Keywords
This article is cited by
-
Improved survival for children and young adolescents with acute myeloid leukemia: a Dutch study on incidence, survival and mortality
Leukemia (2019)
-
The molecular landscape of pediatric acute myeloid leukemia reveals recurrent structural alterations and age-specific mutational interactions
Nature Medicine (2018)
-
Acute Myeloid Leukemia in Children: Experience from Tertiary Cancer Centre in India
Indian Journal of Hematology and Blood Transfusion (2016)
-
Prevalence and prognostic value of IDH1 and IDH2 mutations in childhood AML: a study of the AML–BFM and DCOG study groups
Leukemia (2011)
-
The heterogeneity of pediatric MLL-rearranged acute myeloid leukemia
Leukemia (2011)