Abstract
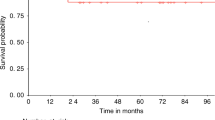
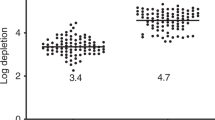
This report examines the impact of graft composition on outcomes in 130 patients with hematological malignancies given unrelated donor granulocyte-colony-stimulating-factor-mobilized peripheral blood mononuclear cells (G-PBMC) (n=116) or marrow (n=14) transplantation after nonmyeloablative conditioning with 90 mg/m2 fludarabine and 2 Gy TBI. The median number of CD34+ cells transplanted was 6.5 × 106/kg. Higher numbers of grafted CD14+ (P=0.0008), CD3+ (P=0.0007), CD4+ (P=0.001), CD8+ (P=0.004), CD3–CD56+ (P=0.003), and CD34+ (P=0.0001) cells were associated with higher levels of day 28 donor T-cell chimerism. Higher numbers of CD14+ (P=0.01) and CD34+ (P=0.0003) cells were associated with rapid achievement of complete donor T-cell chimerism, while high numbers of CD8+ (P=0.005) and CD34+ (P=0.01) cells were associated with low probabilities of graft rejection. When analyses were restricted to G-PBMC recipients, higher numbers of grafted CD34+ cells were associated with higher levels of day 28 donor T-cell chimerism (P=0.01), rapid achievement of complete donor T-cell chimerism (P=0.02), and a trend for lower risk for graft rejection (P=0.14). There were no associations between any cell subsets and acute or chronic GVHD nor relapse/progression. These data suggest more rapid engraftment of donor T cells and reduced rejection rates could be achieved by increasing the doses of CD34+ cells in unrelated grafts administered after nonmyeloablative conditioning.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Girgis M, Hallemeier C, Blum W, Brown R, Lin H-S, Khoury H et al. Chimerism and clinical outcomes of 110 unrelated donor bone marrow transplant recipients conditioned with low dose (550 cGy), single exposure total body irradiation and cyclophosphamide. Blood, prepublished online May 4, 2004; (in press).
Wong R, Giralt SA, Martin T, Couriel DR, Anagnostopoulos A, Hosing C et al. Reduced-intensity conditioning for unrelated donor hematopoietic stem cell transplantation as treatment for myeloid malignancies in patients older than 55 years. Blood 2003; 102: 3052–3059.
Nagler A, Aker M, Or R, Naparstek E, Varadi G, Brautbar C et al. Low-intensity conditioning is sufficient to ensure engraftment in matched unrelated bone marrow transplantation. Exp Hematol 2001; 29: 362–370.
Chakraverty R, Peggs K, Chopra R, Milligan DW, Kottaridis PD, Verfuerth S et al. Limiting transplantation-related mortality following unrelated donor stem cell transplantion by using a nonmyeloablative conditioning regimen. Blood 2002; 99: 1071–1078.
Maris MB, Niederwieser D, Sandmaier BM, Storer B, Stuart M, Maloney D et al. HLA-matched unrelated donor hematopoietic cell transplantation after nonmyeloablative conditioning for patients with hematologic malignancies. Blood 2003; 102: 2021–2030.
Appelbaum FR . Dose intensity and the toxicity and efficacy of allogeneic hematopoietic cell transplantation. Leukemia 2005; 19: 171–175.
Baron F, Maris MB, Sandmaier BM, Storer BE, Sorror M, Diaconescu R et al. Graft-versus-tumor effects after allogeneic hematopoietic cell transplantation with nonmyeloablative conditioning. J Clin Oncol, (in press).
Storb R, Yu C, Wagner JL, Deeg HJ, Nash RA, Kiem H-P et al. Stable mixed hematopoietic chimerism in DLA-identical littermate dogs given sublethal total body irradiation before and pharmacological immunosuppression after marrow transplantation. Blood 1997; 89: 3048–3054.
McSweeney PA, Niederwieser D, Shizuru JA, Sandmaier BM, Molina AJ, Maloney DG et al. Hematopoietic cell transplantation in older patients with hematologic malignancies: replacing high-dose cytotoxic therapy with graft-versus-tumor effects. Blood 2001; 97: 3390–3400.
Niederwieser D, Maris M, Shizuru JA, Petersdorf E, Hegenbart U, Sandmaier BM et al. Low-dose total body irradiation (TBI) and fludarabine followed by hematopoietic cell transplantation (HCT) from HLA-matched or mismatched unrelated donors and postgrafting immunosuppression with cyclosporine and mycophenolate mofetil (MMF) can induce durable complete chimerism and sustained remissions in patients with hematological diseases. Blood 2003; 101: 1620–1629.
Baron F, Maris MB, Storer BE, Sandmaier BM, Stuart MJ, McSweeney PA et al. HLA-matched unrelated donor hematopoietic cell transplantation after nonmyeloablative conditioning for patients with chronic myeloid leukemia. Biol Blood Marrow Transplant, (in press).
Sorror ML, Maris MB, Storer B, Sandmaier BM, Diaconescu R, Flowers C et al. Comparing morbidity and mortality of HLA-matched unrelated donor hematopoietic cell transplantation after nonmyeloablative and myeloablative conditioning: influence of pretransplant comorbidities. Blood 2004; 104: 961–968.
Diaconescu R, Flowers CR, Storer B, Sorror ML, Maris MB, Maloney DG et al. Morbidity and mortality with nonmyeloablative compared to myeloablative conditioning before hematopoietic cell transplantation from HLA matched related donors. Blood 2004; 104: 1550–1558.
Bethge WA, Hegenbart U, Stuart MJ, Storer BE, Maris MB, Flowers MED et al. Adoptive immunotherapy with donor lymphocyte infusions after allogeneic hematopoietic cell transplantation following nonmyeloablative conditioning. Blood 2004; 103: 790–795.
Panse JP, Bastianelli C, Santos EB, Schwarzinger I, Raff RF, Storb R et al. Dog leukocyte antigen nonidentical unrelated canine marrow grafts: enhancement of engraftment by both CD4 and CD8T cells. Transplantation 2003; 76: 474–480.
Ho VT, Soiffer RJ . The history and future of T-cell depletion as graft-versus-host disease prophylaxis for allogeneic hematopoietic stem cell transplantation (Review). Blood 2001; 98: 3192–3204.
Champlin R, Ho W, Gajewski J, Feig S, Burnison M, Holley G et al. Selective depletion of CD8+ T lymphocytes for prevention of graft-versus-host disease after allogeneic bone marrow transplantation. Blood 1990; 76: 418–423.
Baron F, Baker JE, Storb R, Gooley TA, Sandmaier BM, Maris MB et al. Kinetics of engraftment in patients with hematological malignancies given allogeneic hematopoietic cell transplantation after nonmyeloablative conditioning. Blood 2004; 104: 2254–2262.
Cao TM, Shizuru JA, Wong RM, Sheehan K, Laport GG, Stockerl-Goldstein KE et al. Engraftment and survival following reduced-intensity allogeneic peripheral blood hematopoietic cell transplantation is affected by CD8+ T-cell dose. Blood 2005; 105: 2300–2306.
Mielcarek M, Graf L, Johnson G, Torok-Storb B . Production of interleukin-10 by granulocyte colony-stimulating factor-mobilized blood products: a mechanism for monocyte-mediated suppression of T cell proliferation. Blood 1998; 92: 215–222.
Storb R, Prentice RL, Buckner CD, Clift RA, Appelbaum F, Deeg J et al. Graft-versus-host disease and survival in patients with aplastic anemia treated by marrow grafts from HLA-identical siblings. Beneficial effect of a protective environment. N Engl J Med 1983; 308: 302–307.
Blaise D, Kuentz M, Fortanier C, Bourhis JH, Milpied N, Sutton L et al. Randomized trial of bone marrow versus lenograstim-primed blood cell allogeneic transplantation in patients with early-stage leukemia: a report from the Société Française de Greffe de Moelle. J Clin Oncol 2000; 18: 537–571.
Bensinger WI, Martin PJ, Storer B, Clift R, Forman SJ, Negrin R et al. Transplantation of bone marrow as compared with peripheral-blood cells from HLA-identical relatives in patients with hematologic cancers. N Engl J Med 2001; 344: 175–181.
Kernan NA, Collins NM, Juliano L, Cartagenia T, Dupont B, O'Reilly RJ . Clonable T lymphocytes in T cell-depleted bone marrow transplants correlate with development of graft-v-host disease. Blood 1986; 68: 770–773.
Remberger M, Ringdén O, Blau I-W, Ottinger H, Kremens B, Kiehl MG et al. No difference in graft-versus-host disease, relapse, and survival comparing peripheral stem cells to bone marrow using unrelated donors. Blood 2001; 98: 1739–1745.
Zaucha JM, Gooley T, Bensinger WI, Heimfeld S, Chauncey TR, Zaucha R et al. CD34 cell dose in granulocyte colony-stimulating factor-mobilized peripheral blood mononuclear cell grafts affects engraftment kinetics and development of extensive chronic graft-versus-host disease after human leukocyte antigen-identical sibling transplantation. Blood 2001; 98: 3221–3227.
Mohty M, Bilger K, Jourdan E, Kuentz M, Michallet M, Bourhis JH et al. Higher doses of CD34+ peripheral blood stem cells are associated with increased mortality from chronic graft-versus-host disease after allogeneic HLA-identical sibling transplantation. Leukemia 2003; 17: 869–875.
Perez-Simon JA, Diez-Campelo M, Martino R, Sureda A, Caballero D, Canizo C et al. Impact of CD34+ cell dose on the outcome of patients undergoing reduced-intensity-conditioning allogeneic peripheral blood stem cell transplantation. Blood 2003; 102: 1108–1113.
Riddell SR, Berger C, Murata M, Randolph S, Warren EH . The graft versus leukemia response after allogeneic hematopoietic stem cell transplantation. Blood Rev 2003; 17: 153–162.
Panse JP, Heimfeld S, Guthrie KA, Maris MB, Maloney DG, Baril BB et al. Allogeneic peripheral blood stem cell graft composition affects early T-cell chimerism and later clinical outcomes after nonmyeloablative conditioning. Br J Haematol 2005; 128: 659–667.
Sierra J, Storer B, Hansen JA, Bjerke JW, Martin PJ, Petersdorf EW et al. Transplantation of marrow cells from unrelated donors for treatment of high-risk acute leukemia: the effect of leukemic burden, donor HLA-matching, and marrow cell dose. Blood 1997; 89: 4226–4235.
Heimfeld S . Bone marrow transplantation: how important is CD34 cell dose in HLA-identical stem cell transplantation (Keynote Address). Leukemia 2003; 17: 856–858.
Acknowledgements
We thank the data coordinators Heather Hildebrant Debbie Bassuk, and Chris Davis, and the study nurses Steve Minor, Mary Hinds, and John Sedgwick for their invaluable help in making the study possible. We also wish to thank Bonnie Larson and Helen Crawford for help with manuscript preparation, and the physicians, nurses, and support personnel for their care of patients involved in this study. This work was supported in part by Grants CA78902, CA92058, HL36444, CA18029, HD44175, DK56465, and CA15704 from the National Institutes of Health, Department of Health and Human Services (DHHS), Bethesda, MD. DGM was supported by a grant from the Gabrielle Rich Leukemia Foundation, and MS by a grant from the Oncology Research Faculty Development Program of the NCI. RS also received support from the Laura Landro Salomon Endowment Fund. FB is research associate of the National Fund for Scientific Research (FNRS) Belgium and supported in part by postdoctoral grants from the Fulbright Commission and from the Centre Anticancéreux près l'Université de Liège.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baron, F., Maris, M., Storer, B. et al. High doses of transplanted CD34+ cells are associated with rapid T-cell engraftment and lessened risk of graft rejection, but not more graft-versus-host disease after nonmyeloablative conditioning and unrelated hematopoietic cell transplantation. Leukemia 19, 822–828 (2005). https://doi.org/10.1038/sj.leu.2403718
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403718
Keywords
This article is cited by
-
Reduced-intensity conditioned allogeneic SCT in adults with AML
Bone Marrow Transplantation (2015)
-
Low blood lymphocyte count at 30 days post transplant predicts worse acute GVHD and survival but not relapse in a large retrospective cohort
Bone Marrow Transplantation (2015)
-
Biologic markers of chronic GVHD
Bone Marrow Transplantation (2014)
-
Extensive chronic GVHD is associated with donor blood CD34+ cell count after G-CSF mobilization in non-myeloablative allogeneic PBSC transplantation
Bone Marrow Transplantation (2012)
-
Salvage haploidentical transplantation for graft failure using reduced-intensity conditioning
Bone Marrow Transplantation (2012)