Abstract
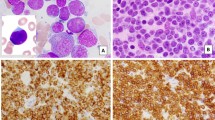
Aggressive natural killer-cell leukemia (ANKL) is a rare form of large granular lymphocyte leukemia, which is characterized by a systemic proliferation of NK cells. The clinical features of 22 ANKL cases were analyzed. Hepatomegaly (64%), splenomegaly (55%) and lymphadenopathy (41%) were also frequently observed. Leukemic cells were identified as CD1−, CD2+, surface CD3−, CD4−, CD5−, CD7+, CD8+/−, CD10−, CD11b+/−, CD13−, CD16+, CD19−, CD20−, CD25−, CD33−, CD34−, CD38+, CD56+, CD122+, HLA-DR+ and TCR−. Two of the 16 cases examined for CD57 were positive and three of the seven cases examined for cytoplasmic CD3. Epstein–Barr virus was detected in the tumor cells of 11 of the 13 cases examined. No common cytogenetic abnormalities were identified and 6q anomaly was detected in only one. Three of 13 patients treated with chemotherapy containing anthracycline/anthraquinone attained complete remission, in contrast to none of the eight who were treated with regimens without anthracycline. Although the overall prognosis was poor with a median survival of 58 days, those who attained remission showed better prognosis (P=0.005). These findings suggest that ANKL is an entity of mature cytotoxic NK-cell neoplasms with distinct phenotype and disease presentations. Intensive treatment for ANKL may result in a better prognosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Chan JKC, Wong KF, Jaffe ES, Ralfkiaer E . Aggressive NK-cell leukaemia. In: Jaffe ES, Harris NL, Stein H, Vardiman JW (eds). World Health Organization Classification of Tumours: Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. Lyon: IARC Press, 2001, pp 198–200.
Imamura N, Kusunoki Y, Kawa-Ha K, Yumura K, Hara J, Oda K et al. Aggressive natural killer cell leukaemia/lymphoma: report of four cases and review of the literature. Br J Haematol 1990; 75: 49–59.
Loughran Jr TP . Clonal diseases of large granular lymphocytes. Blood 1993; 82: 1–14.
Jaffe ES . Classification of natural killer (NK) cell and NK-like T-cell malignancies. Blood 1996; 15: 1207–1210.
Oshimi K . Lymphoproliferative disorders of natural killer cells. Int J Hematol 1996; 63: 279–290.
Yodoi J, Teshigawara K, Nikaido T, Fukui K, Noma T, Honjo T et al. TCGF (IL 2)-receptor inducing factor(s). I. Regulation of IL 2 receptor on a natural killer-like cell line (YT cells). J Immunol 1985; 134: 1623–1630.
Fernandez LA, Pope B, Lee C, Zayed E . Aggressive natural killer cell leukemia in an adult and establishment of an NK cell line. Blood 1986; 67: 925–930.
Koizumi S, Seki H, Tachinami T, Taniguchi M, Matsuda A, Taga K et al. Malignant clonal expansion of large granular lymphocytes with a Leu-11+, Leu-7− surface phenotype: in vitro responsiveness of malignant cells to recombinant human interleukin 2. Blood 1986; 68: 1065–1073.
Kawa-Ha K, Ishihara S, Ninomiya T, Yumura-Yagi K, Hara J, Murayama F et al. CD3-negative lymphoproliferative disease of granular lymphocytes containing Epstein–Barr viral DNA. J Clin Invest 1989; 84: 51–55.
Taniwaki M, Tagawa S, Nishigaki H, Horiike S, Misawa S, Shimazaki C et al. Chromosomal abnormalities define clonal proliferation in CD3- large granular lymphocyte leukemia. Am J Hematol 1990; 33: 32–38.
Oshimi K, Yamada O, Kaneko T, Nishinarita S, Iizuka Y, Urabe A et al. Laboratory findings and clinical courses of 33 patients with granular lymphocyte-proliferative disorders. Leukemia 1993; 7: 782–788.
Kwong YL, Wong KF, Chan LC, Liang RH, Chan JK, Lin CK et al. Large granular lymphocyte leukemia. A study of nine cases in a Chinese population. Am J Clin Pathol 1995; 103: 76–81.
Shimodaira S, Ishida F, Kobayashi H, Mahbub B, Kawa-Ha K, Kitano K . The detection of clonal proliferation in granular lymphocyte-proliferative disorders of natural killer cell lineage. Br J Haematol 1995; 90: 578–584.
Chan JK, Sin VC, Wong KF, Ng CS, Tsang WY, Chan CH et al. Nonnasal lymphoma expressing the natural killer cell marker CD56: a clinicopathologic study of 49 cases of an uncommon aggressive neoplasm. Blood 1997; 89: 4501–4513.
Wong KF, Chan JK, Kwong YL . Identification of del(6)(q21q25) as a recurring chromosomal abnormality in putative NK cell lymphoma/leukaemia. Br J Haematol 1997; 98: 922–926.
Mizuno S, Akashi K, Ohshima K, Iwasaki H, Miyamoto T, Uchida N et al. Interferon-gamma prevents apoptosis in Epstein–Barr virus-infected natural killer cell leukemia in an autocrine fashion. Blood 1999; 93: 3494–3504.
Akashi K, Mizuno S . Epstein–Barr virus-infected natural killer cell leukemia. Leukemia Lymphoma 2000; 40: 57–66.
Sheridan W, Winton EF, Chan WC, Gordon DS, Vogler WR, Phillips C et al. Leukemia of non-T lineage natural killer cells. Blood 1988; 72: 1701–1707.
Ohno T, Kanoh T, Arita Y, Fujii H, Kuribayashi K, Masuda T et al. Fulminant clonal expansion of large granular lymphocytes. Characterization of their morphology, phenotype, genotype, and function. Cancer 1988; 62: 1918–1927.
Ohno Y, Amakawa R, Fukuhara S, Huang CR, Kamesaki H, Amano H et al. Acute transformation of chronic large granular lymphocyte leukemia associated with additional chromosome abnormality. Cancer 1989; 64: 63–67.
Lin CK, Liu HW, Tse PW, Lai CL, Chan GT . A patient with large granular lymphocytosis of unusual phenotype and polymorphic T-cell receptor beta-chain gene rearrangement. Am J Clin Pathol 1990; 94: 211–216.
Okuda T, Sakamoto S, Deguchi T, Misawa S, Kashima K, Yoshihara T et al. Hemophagocytic syndrome associated with aggressive natural killer cell leukemia. Am J Hematol 1991; 38: 321–323.
Imashuku S, Okuda T, Yoshihara T, Ikushima S, Hibi S . Cytokine levels in aggressive natural killer cell leukemia and malignant histiocytosis. Br J Haematol 1991; 79: 132–133.
Tsukada T, Ohno T, Morita K, Otsuji H, Sekine T, Nishikawa M et al. Aggressive natural killer cell lymphoproliferative disease of large granular lymphocytes with leukemia-like clinical course in the terminal stage. Rinsho Ketsueki 1991; 32: 162–166.
Hart DNJ, Baker BW, Inglis MJ, Nimmo JC, Starling GC, Deacon E et al. Epstein–Barr viral DNA in acute large granular lymphocyte (natural killer) leukemic cells. Blood 1992; 79: 2116–2123.
Sun T, Schulman P, Kolitz J, Susin M, Brody J, Koduru P et al. A study of lymphoma of large granular lymphocytes with modern modalities: report of two cases and review of the literature. Am J Hematol 1992; 40: 135–145.
Kojima H, Suzukawa K, Yatabe Y, Hori M, Nagasawa T, Abe T . Establishment of a new natural killer (NK) cell line, TKS-1, from a patient with aggressive type of large granular lymphocyte (LGL) leukemia. Leukemia 1994; 8: 1999–2004.
Gelb AB, van de Rijn M, Regula Jr DP, Regula Jr DP, Cornbleet JP, Kamel OW et al. Epstein–Barr virus-associated natural killer-large granular lymphocyte leukemia. Hum Pathol 1994; 25: 953–960.
Nichols GE, Normansell DE, Williams ME . Lymphoproliferative disorder of granular lymphocytes: nine cases including one with features of CD56 (NKH1)-positive aggressive natural killer cell lymphoma. Mod Pathol 1994; 7: 819–824.
Yatabe Y, Mori N, Hirabayashi N, Asai J . Natural killer cell leukemia. An autopsy case. Arch Pathol Lab Med 1994; 118: 1201–1204.
Robertson MJ, Cochran KJ, Cameron C, Le JM, Tantravahi R, Ritz J . Characterization of a cell line, NKL, derived from an aggressive human natural killer cell leukemia. Exp Hematol 1996; 24: 406–415.
Teshima T, Miyaji R, Fukuda M, Ohshima K . Bone-marrow transplantation for Epstein–Barr-virus-associated natural killer cell-large granular lymphocyte leukaemia. Lancet 1996; 347: 1124.
Takami A, Nakao S, Yachie A, Kasahara Y, Okumura H, Miura Y et al. Successful treatment of Epstein–Barr virus-associated natural killer cell large granular lymphocytic leukaemia using allogeneic peripheral blood stem cell transplantation. Bone Marrow Transplant 1998; 21: 1279–1282.
Mateu R, Badell I, Alcala A, Estivill C, Rubiol E, Nomdedeu J et al. Aggressive natural killer cell leukemia: report of a case in a Caucasian boy. Haematologica 1998; 83: 190–192.
Dunkley S, Gibson J, Mackinlay N, Joshua D . Aggressive natural killer cell leukemia/lymphoma. Pathology 1998; 30: 157–159.
Yagita M, Huang CL, Umehara H, Matsuo Y, Tabata R, Miyake M et al. A novel natural killer cell line (KHYG-1) from a patient with aggressive natural killer cell leukemia carrying a p53 point mutation. Leukemia 2000; 14: 922–930.
Kuroda J, Kimura S, Akaogi T, Hayashi H, Nishida K, Kakazu N et al. Aggressive natural killer cell leukemia/lymphoma: a comprehensive cytogenetic study by spectral karyotyping. Ann Hematol 2000; 79: 519–522.
Allory Y, Challine D, Haioun C, Copie-Bergman C, Delfau-Larue MH, Boucher E et al. Bone marrow involvement in lymphomas with hemophagocytic syndrome at presentation: a clinicopathologic study of 11 patients in a Western institution. Am J Surg Pathol 2001; 25: 865–874.
Yamashiro T, Watanabe N, Yokoyama K, Koga C, Tsuruo T, Kobayashi Y . Requirement of expression of P-glycoprotein on human natural killer leukemia cells for cell-mediated cytotoxicity. Biochem Pharmacol 1998; 55: 1385–1390.
Egashira M, Kawamata N, Sugimoto K, Kaneko T, Oshimi K . P-glycoprotein expression on normal and abnormally expanded natural killer cells and inhibition of P-glycoprotein function by cyclosporin A and its analogue, PSC833. Blood 1999; 93: 599–606.
Song SY, Kim WS, Ko YH, Kim K, Lee MH, Park K . Aggressive natural killer cell leukemia; clinical features and treatment outcome. Haematologica 2002; 87: 1343–1345.
Oshimi K . Leukemia and lymphoma of natural killer lineage cells. Int J Hematol 2003; 78: 18–23.
Suzuki R, Yamamoto K, Seto M, Kagami Y, Ogura M, Yatabe Y et al. CD7+ and CD56+ myeloid/NK cell precursor acute leukemia: a distinct hematolymphoid disease entity. Blood 1997; 90: 2417–2428.
Brousset P, Butet V, Chittal S, Selves J, Delsol G . Comparison of in situ hybridization using different nonisotopic probes for detection of Epstein–Barr virus in nasopharyngeal carcinoma and immunohistochemical correlation with anti-latent membrane protein antibody. Lab Invest 1992; 67: 457–464.
Sasajima Y, Yamabe H, Kobashi Y, Hirai K, Mori S . High expression of the Epstein–Barr virus latent protein EB nuclear antigen-2 on pyothorax-associated lymphomas. Am J Pathol 1993; 143: 1280–1285.
The International Non-Hodgkin's Lymphoma Prognostic Factors Project. A predictive model for aggressive non-Hodgkin's lymphoma. N Engl J Med 1993; 329: 987–994.
Soler J, Bordes R, Ortuno F, Montagud M, Martorell J, Pons C et al. Aggressive natural killer cell leukaemia/lymphoma in two patients with lethal midline granuloma. Br J Haematol 1994; 86: 659–662.
Quintanilla-Martinez L, Jaffe ES . Aggressive NK cell lymphomas: insights into the spectrum of NK cell derived malignancies. Histopathology 2000; 37: 372–374.
Procopio AD, Paolini R, Vecchio I, Frati L, Santoni A . GTP-binding proteins transduce signals generated via human FC gamma receptor IIIA (CD16). J Immunol 1991; 146: 3550–3556.
Kaneko T, Fukuda J, Yoshihara T, Zheng H, Mori S, Mizoguchi H et al. Nasal natural killer (NK) cell lymphoma: report of a case with activated NK cells containing Epstein–Barr virus and expressing CD21 antigen, and comparative studies of their phenotype and cytotoxicity with normal NK cells. Br J Haematol 1995; 91: 355–361.
Lanier LL, Le AM, Civin CI, Loken MR, Phillips JH . The relationship of CD16 (Leu-11) and Leu-19 (NKH-1) antigen expression on human peripheral blood NK cells and cytotoxic lymphocytes. J Immunol 1986; 136: 4480–4486.
Cooper MA, Fehniger TA, Turner SC, Chen KS, Ghaheri BA, Ghayur T et al. Human natural killer cells: a unique innate immunoregulatory role for the CD56bright subset. Blood 2001; 97: 3146–3151.
Farag SS, VanDeusen JB, Feninger TA, Caligiuri MA . Biology and clinical impact of human natural killer cells. Int J Hematol 2003; 78: 7–17.
Caligiuri MA, Zmuidzinas A, Manley TJ, Levine H, Smith KA, Rits J . Functional consequences of interleukin-2 receptor expression on resting human lymphocytes: identification of a novel natural killer cell subset with high affinity receptors. J Exp Med 1990; 171: 1509–1526.
Weidmann E . Hepatosplenic T cell lymphoma. A review on 45 cases since the first report describing the disease as a distinct lymphoma entity in 1990. Leukemia 2000; 14: 991–997.
Macon WR, Levy NB, Kurtin PJ, Salhany KE, Elkhalifa MY, Casey TT et al. Hepatosplenic αβ T-cell lymphomas: a report of 14 cases and comparison with hepatosplenic γδ T-cell lymphomas. Am J Surg Pathol 2001; 25: 285–296.
Tien HF, Su IJ, Tang JL, Liu MC, Lee FY, Chen YC et al. Clonal chromosomal abnormalities as direct evidence for clonality in nasal T/natural killer cell lymphomas. Br J Haematol 1997; 97: 621–625.
Siu LL, Wong KF, Chan JK, Kwong YL . Comparative genomic hybridization analysis of natural killer cell lymphoma/leukemia. Recognition of consistent patterns of genetic alterations. Am J Pathol 1999; 155: 1419–1425.
Siu LL, Chan V, Chan JK, Wong KF, Liang R, Kwong YL . Consistent patterns of allelic loss in natural killer cell lymphoma. Am J Pathol 2000; 157: 1803–1809.
Sakajiri S, Kawamata N, Egashira M, Mori K, Oshimi K . Molecular analysis of tumor suppressor genes, Rb, p53, p16INK4A, p15INK4B and p14ARF in natural killer cell neoplasms. Jpn J Cancer Res 2001; 92: 1048–1056.
Oka T, Yoshino T, Hayashi K, Ohara N, Nakanishi T, Yamaai Y et al. Reduction of hematopoietic cell-specific tyrosine phosphatase SHP-1 gene expression in natural killer cell lymphoma and various types of lymphomas/leukemias: combination analysis with cDNA expression array and tissue microarray. Am J Pathol 2001; 159: 1495–1505.
Trambas C, Wang Z, Cianfriglia M, Woods G . Evidence that natural killer cells express mini P-glycoproteins but not classic 170 kDa P-glycoprotein. Br J Haematol 2001; 114: 177–184.
Nagafuji K, Fujisaki T, Arima F, Ohshima K . L-asparaginase induced durable remission of relapsed nasal NK/T-cell lymphoma after autologous peripheral blood stem cell transplantation. Int J Hematol 2001; 74: 447–450.
Yong W, Zheng W, Zhang Y . Clinical characteristics and treatment of midline nasal and nasal type NK/T cell lymphoma. Zhonghua Yi Xue Za Zhi 2001; 81: 773–775.
Acknowledgements
This work was supported in part by a grant-in-aid for the second-term comprehensive 10-year strategy for cancer control from the Ministry of Health and Welfare, a grant-in-aid for science on primary areas (Cancer Research), from the Ministry of Education, Science and Culture, Japan. We thank the collaborators from the following institutions for providing the patients' data and specimens: Saiseikai Maebashi Hospital; Mie University; Japanese Red Cross Nagoya First Hospital; Kitano Hospital; Dokkyo University School of Medicine; Fujita Health University School of Medicine; Hiroshima University School of Medicine; Kansai Medical University; Toride Kyodo Hospital; Tokyo Medical and Dental University; Kyushu University and a grant-in-aid for Encouragement of Young Scientists from the Ministry of Education, Science and Culture, Japan.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Suzuki, R., Suzumiya, J., Nakamura, S. et al. Aggressive natural killer-cell leukemia revisited: large granular lymphocyte leukemia of cytotoxic NK cells. Leukemia 18, 763–770 (2004). https://doi.org/10.1038/sj.leu.2403262
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403262
Keywords
This article is cited by
-
Initial diagnosis of extranodal NK/T-cell lymphoma in pericardial fluid with concomitant hemophagocytic lymphohistiocytosis (HLH)
Journal of Hematopathology (2024)
-
A case of acute liver failure due to aggressive natural killer-cell leukemia with a rapid course
Clinical Journal of Gastroenterology (2023)
-
Expanding anti-CD38 immunotherapy for lymphoid malignancies
Journal of Experimental & Clinical Cancer Research (2022)
-
Allogeneic stem cell transplantation for patients with aggressive NK-cell leukemia
Bone Marrow Transplantation (2021)
-
Correlation of CD38 expression with the progression of hemorrhagic fever with renal syndrome
Archives of Virology (2021)