Abstract
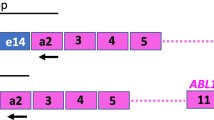
The fusion transcript AML1/ETO corresponding to translocation t(8;21)(q22;q22) can be found in approximately 7–12% of childhood de novo AML. Despite the favorable prognosis, some of these patients relapse. Most of MRD studies so far were performed on adults treated not uniformly. Therefore, we analyzed the follow-up of 15 AML1/ETO-positive children using real-time quantitative reverse transcription PCR (RQ-RT-PCR), all enrolled in the multicenter therapy trial AML-BFM 98. AML1/ETO copy numbers were normalized to the control gene ABL and the results were expressed in copy numbers AML1/ETO per 10 000 copies ABL. At diagnosis, a median of 10 789 copies AML1/ETO was found. A linear decrease to about 10 copies (2–4 log) could be seen in most of the children by the start of consolidation. In the majority of cases they remained positive at this low level during the ongoing therapy. Four children relapsed and two of them had a decrease of less than 2 log before starting consolidation. Three of the relapsed children showed, prior to relapse, an increase of the AML1/ETO fusion transcript at 6, 9, and 11 weeks, respectively. These results suggest that monitoring of minimal residual disease using RQ-RT-PCR could be helpful in detecting patients with a higher risk of relapse.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Erickson P, Gao J, Chang KS, Look T, Whisenant E, Raimondi S et al. Identification of breakpoints in t(8;21) acute myelogenous leukemia and isolation of a fusion transcript, AML1/ETO, with similarity to Drosophila segmentation gene, runt. Blood 1992; 80: 1825–1831.
Miyoshi H, Kozu T, Shimizu K, Enomoto K, Maseki N, Kaneko Y et al. The t(8;21) translocation in acute myeloid leukemia results in production of an AML1-MTG8 fusion transcript. EMBO J 1993; 12: 2715–2721.
Wolford JK, Prochazka M . Structure and expression of the human MTG8/ETO gene. Gene 1998; 212: 103–109.
Tighe JE, Daga A, Calabi F . Translocation breakpoints are clustered on both chromosome 8 and chromosome 21 in the t(8;21) of acute myeloid leukemia. Blood 1993; 81: 592–596.
Kozu T, Miyoshi H, Shimizu K, Maseki N, Kaneko Y, Asou H et al. Junctions of the AML1/MTG8(ETO) fusion are constant in t(8;21) acute myeloid leukemia detected by reverse transcription polymerase chain reaction. Blood 1993; 82: 1270–1276.
van Dongen JJ, Macintyre EA, Gabert JA, Delabesse E, Rossi V, Saglio G et al. Standardized RT-PCR analysis of fusion gene transcripts from chromosome aberrations in acute leukemia for detection of minimal residual disease. Report of the BIOMED-1 Concerted Action: investigation of minimal residual disease in acute leukemia. Leukemia 1999; 13: 1901–1928.
Saunders MJ, Tobal K, Keeney S, Liu YJ . Expression of diverse AML1/MTG8 transcripts is a consistent feature in acute myeloid leukemia with t(8;21) irrespective of disease phase. Leukemia 1996; 10: 1139–1142.
van de Locht LT, Smetsers TF, Wittebol S, Raymakers RA, Mensink EJ . Molecular diversity in AML1/ETO fusion transcripts in patients with t(8;21) positive acute myeloid leukaemia. Leukemia 1994; 8: 1780–1784.
Tighe JE, Calabi F . Alternative, out-of-frame runt/MTG8 transcripts are encoded by the derivative (8) chromosome in the t(8;21) of acute myeloid leukemia M2. Blood 1994; 84: 2115–2121.
Grimwade D, Walker H, Oliver F, Wheatley K, Harrison C, Harrison G et al. The importance of diagnostic cytogenetics on outcome in AML: analysis of 1612 patients entered into the MRC AML 10 trial. The Medical Research Council Adult and Children's Leukaemia Working Parties. Blood 1998; 92: 2322–2333.
Gallego M, Carroll AJ, Gad GS, Pappo A, Head D, Behm F et al. Variant t(8;21) rearrangements in acute myeloblastic leukemia of childhood. Cancer Genet Cytogenet 1994; 75: 139–144.
Andrieu V, Radford-Weiss I, Troussard X, Chane C, Valensi F, Guesnu M et al. Molecular detection of t(8;21)/AML1-ETO in AML M1/M2: correlation with cytogenetics, morphology and immunophenotype. Br J Haematol 1996; 92: 855–865.
Jurlander J, Caligiuri MA, Ruutu T, Baer MR, Strout MP, Oberkircher AR et al. Persistence of the AML1/ETO fusion transcript in patients treated with allogeneic bone marrow transplantation for t(8;21) leukemia. Blood 1996; 88: 2183–2191.
Kusec R, Laczika K, Knobl P, Friedl J, Greinix H, Kahls P et al. AML1/ETO fusion mRNA can be detected in remission blood samples of all patients with t(8;21) acute myeloid leukemia after chemotherapy or autologous bone marrow transplantation. Leukemia 1994; 8: 735–739.
Nucifora G, Larson RA, Rowley JD . Persistence of the 8;21 translocation in patients with acute myeloid leukemia type M2 in long-term remission. Blood 1993; 82: 712–715.
Sakata N, Okamura T, Inoue M, Yumura-Yagi K, Hara J, Tawa A et al. Rapid disappearance of AML1/ETO fusion transcripts in patients with t(8;21) acute myeloid leukemia following bone marrow transplantation and chemotherapy. Leukemia Lymphoma 1997; 26: 141–152.
Satake N, Maseki N, Kozu T, Sakashita A, Kobayashi H, Sakurai M et al. Disappearance of AML1-MTG8 (ETO) fusion transcript in acute myeloid leukaemia patients with t(8;21) in long-term remission. Br J Haematol 1995; 91: 892–898.
Preudhomme C, Philippe N, Macintyre E, Henic N, Lai JL, Jouet JP et al. Persistence of AML1/ETO fusion mRNA in t(8;21) acute myeloid leukemia (AML) in prolonged remission: is there a consensus? [letter]. Leukemia 1996; 10: 186–188.
Morschhauser F, Cayuela JM, Martini S, Baruchel A, Rousselot P, Socie G et al. Evaluation of minimal residual disease using reverse-transcription polymerase chain reaction in t(8;21) acute myeloid leukemia: a multicenter study of 51 patients. J Clin Oncol 2000; 18: 788–794.
Muto A, Mori S, Matsushita H, Awaya N, Ueno H, Takayama N et al. Serial quantification of minimal residual disease of t(8;21) acute myelogenous leukaemia with RT-competitive PCR assay. Br J Haematol 1996; 95: 85–94.
Tobal K, Newton J, Macheta M, Chang J, Morgenstem G, Evans PA et al. Molecular quantitation of minimal residual disease in acute myeloid leukemia with t(8;21) can identify patients in durable remission and predict clinical relapse. Blood 2000; 95: 815–819.
Tobal K, Yin JA . Monitoring of minimal residual disease by quantitative reverse transcriptase-polymerase chain reaction for AML1-MTG8 transcripts in AML-M2 with t(8; 21). Blood 1996; 88: 3704–3709.
Wattjes MP, Krauter J, Nagel S, Heidenreich O, Ganser A, Heil G . Comparison of nested competitive RT-PCR and real-time RT-PCR for the detection and quantification of AML1/MTG8 fusion transcripts in t(8;21) positive acute myelogenous leukemia. Leukemia 2000; 14: 329–335.
Kondo M, Kudo K, Kimura H, Inaba J, Kato K, Kojima S et al. Real-time quantitative reverse transcription-polymerase chain reaction for the detection of AML1-MTG8 fusion transcripts in t(8;21)-positive acute myelogenous leukemia. Leukemia Res 2000; 24: 951–956.
Creutzig U, Ritter J, Zimmermann M, Reinhardt D, Hermann J, Berthold F et al. Improved treatment results in high-risk pediatric acute myeloid leukemia patients after intensification with high-dose cytarabine and mitoxantrone: results of Study Acute Myeloid Leukemia-Berlin–Frankfurt–Munster 93. J Clin Oncol 2001; 19: 2705–2713.
Maruyama F, Stass SA, Estey EH, Cork A, Hirano M, Ino T et al. Detection of AML1/ETO fusion transcript as a tool for diagnosing t(8;21) positive acute myelogenous leukemia. Leukemia 1994; 8: 40–45.
Hochhaus A, Lin F, Reiter A, Skladny H, Mason PJ, van RF et al. Quantification of residual disease in chronic myelogenous leukemia patients on interferon-alpha therapy by competitive polymerase chain reaction. Blood 1996; 87: 1549–1555.
Beillard E, Pallisgaard N, Bi W, van der Velden V, Dee R, van der Schoot E et al. Evaluation of candidate control genes for diagnosis and MRD detection in leukemic patients using ‘real-time’ quantitative reverse-transcriptase polymerase chain reaction (RQ-PCR) – A Europe Against Cancer Program. Leukemia (in press).
Asou H, Tashiro S, Hamamoto K, Otsuji A, Kita K, Kamada N . Establishment of a human acute myeloid leukemia cell line (Kasumi-1) with 8;21 chromosome translocation. Blood 1991; 77: 2031–2036.
Drexler HG, Dirks W, MacLeod RAF, Quentmeier H, Steube K, Uphoff CC . Human and animal cell lines. DSMZ Cat 1997, 6th edn, pp 75--84.
Gabert J, Beillard E, van der Velden V, Bi W, Grimwade D, Pallisgaard N et al. Standardization and quality control studies of ‘real-time’ quantitative reverse transcriptase polymerase chain reaction (RQ-PCR) of fusion gene transcripts for residual disease detection in leukemia – A Europe Against Cancer Program. Leukemia (in press).
Lion T . Monitoring of residual disease in chronic myelogenous leukemia by quantitative polymerase chain reaction and clinical decision making. Blood 1999; 94: 1486–1488.
Sugimoto T, Das H, Imoto S, Murayama T, Gomyo H, Chakraborty S et al. Quantitation of minimal residual disease in t(8;21)-positive acute myelogenous leukemia patients using real-time quantitative RT-PCR. Am J Hematol 2000; 64: 101–106.
Fujimaki S, Funato T, Harigae H, Imaizumi M, Suzuki H, Kaneko Y et al. A quantitative reverse transcriptase polymerase chain reaction method for the detection of leukaemic cells with t(8;21) in peripheral blood. Eur J Haematol 2000; 64: 252–258.
Krauter J, Wattjes MP, Nagel S, Heidenreich O, Krug U, Kafert S et al. Real-time RT-PCR for the detection and quantification of AML1/MTG8 fusion transcripts in t(8;21)-positive AML patients. Br J Haematol 1999; 107: 80–85.
Marcucci G, Livak KJ, Bi W, Strout MP, Bloomfield CD, Caligiuri MA . Detection of minimal residual disease in patients with AML1/ETO-associated acute myeloid leukemia using a novel quantitative reverse transcription polymerase chain reaction assay. Leukemia 1998; 12: 1482–1489.
Barragan E, Bolufer P, Moreno I, Martin G, Nomdedeu J, Brunet S et al. Quantitative detection of AML1 – ETO rearrangement by real-time RT-PCR using fluorescently labeled probes. Leukemia Lymphoma 2001; 42: 747–756.
Krauter J, Heil G, Ganser A . The AML1/MTG8 fusion transcript in t(8;21) positive AML and its implication for the detection of minimal residual disease. Hematol 2001; 5: 369–381.
Krauter J, Hoellge W, Wattjes MP, Nagel S, Heidenreich O, Bunjes D et al. Detection and quantification of CBFB/MYH11 fusion transcripts in patients with inv(16)-positive acute myeloblastic leukemia by real-time RT-PCR. Genes Chromosomes Cancer 2001; 30: 342–348.
Pallisgaard N, Clausen N, Schroder H, Hokland P . Rapid and sensitive minimal residual disease detection in acute leukemia by quantitative real-time RT-PCR exemplified by t(12;21) TEL–AML1 fusion transcript. Genes Chromosomes Cancer 1999; 26: 355–365.
Guerrasio A, Pilatrino C, De Micheli D, Cilloni D, Serra A, Gottardi E et al. Assessment of minimal residual disease (MRD) in CBFbeta/MYH 11-positive acute myeloid leukemias by qualitative and quantitative RT-PCR amplification of fusion transcripts. Leukemia 2002; 16: 1176–1181.
Buonamici S, Ottaviani E, Testoni N, Montefusco V, Visani G, Bonifazi F et al. Real-time quantitation of minimal residual disease in inv(16)-positive acute myeloid leukemia may indicate risk for clinical relapse and may identify patients in a curable state. Blood 2002; 99: 443–449.
Acknowledgements
This work was supported by the German Kinderkrebsstiftung, the Parents' Initiative Gießen, the Kompetenznetzwerk Pädiatrische Hämatologie und Onkologie (Grant No. 01G19963/2), and the SANCO European Commission within the Europe Against Cancer Program (EAC). We thank the technical staff of the Oncogenetic Laboratory, especially Yvonne Schichowski, for excellent assistance.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Viehmann, S., Teigler-Schlegel, A., Bruch, J. et al. Monitoring of minimal residual disease (MRD) by real-time quantitative reverse transcription PCR (RQ-RT-PCR) in childhood acute myeloid leukemia with AML1/ETO rearrangement. Leukemia 17, 1130–1136 (2003). https://doi.org/10.1038/sj.leu.2402959
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402959
Keywords
This article is cited by
-
Methods of Detection of Measurable Residual Disease in AML
Current Hematologic Malignancy Reports (2017)
-
BAALC expression: a suitable marker for prognostic risk stratification and detection of residual disease in cytogenetically normal acute myeloid leukemia
Blood Cancer Journal (2014)
-
Overview of Therapy and Strategies for Optimizing Outcomes in De Novo Pediatric Acute Myeloid Leukemia
Pediatric Drugs (2014)
-
Progress of Minimal Residual Disease Studies in Childhood Acute Leukemia
Current Hematologic Malignancy Reports (2010)
-
Abschlusssymposium des Kompetenznetzes Pädiatrische Onkologie und Hämatologie
Der Onkologe (2008)