Abstract
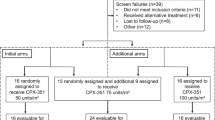
HuM195 is a humanized, unconjugated, anti-CD33 monoclonal antibody. Fifty adult patients with relapsed or refractory AML were randomized to receive HuM195 at a dose of 12 or 36 mg/m2 by intravenous infusion over 4 h on days 1–4 and 15–18. Patients with stable or responding disease received two additional cycles on days 29–32 and 43–46. HuM195 was given as first salvage therapy in 24 patients and as second or subsequent salvage therapy in 26 patients. Pretreatment blast percentage in the marrow was between 5 and 30% in 20 patients with the others having blast counts greater than 30%. The median age of patients was 62 years (range 26–86) and CD33 was detected in 95% of patients for whom immunophenotyping was available. Of 49 evaluable patients, two complete and one partial remission were observed. All three responses were in patients treated at the 12 mg/m2 dose level and all had baseline blast percentages less than 30%. Decreases in blast counts ranging from 30 to 74% were seen in nine additional patients. Infusion-related events of fever and chills occurred in the majority of patients and were generally mild and primarily related to the first dose of antibody. No hepatic, renal or cardiac toxicities were observed and other adverse events such as nausea, vomiting, mucositis and diarrhea were uncommon or felt to be unrelated to HuM195. In addition, anti-HuM195 responses were not detected. HuM195 as a single agent has minimal, but observable, anti-leukemic activity in patients with relapsed or refractory AML and activity is confined to patients with low burden disease. No significant differences in clinical efficacy or toxicity were seen between the two dose levels of antibody. HuM195 was well tolerated with infusion-related fevers and chills the predominant toxicities seen. Meaningful clinical efficacy of this unconjugated monoclonal antibody may be realized only in patients with minimal residual disease, or in combination with chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Zittoun R . Chemotherapy of acute myelogenous leukemia. A review. Leukemia 1992; 6 (Suppl. 2), 36–38.
Mrozek K, Heinonen K, de la Chapelle A, Bloomfield C . Clinical significance of cytogenetics in acute myeloid leukemia. Semin Oncol 1997; 24: 17–31.
Estey E, Thall P, David C . Design and analysis of trials of salvage therapy in acute myelogenous leukemia. Cancer Chemother Pharmacol 1997; l 40 (Suppl.): S9–12.
Gajewski JL, Ho WG, Nimer SD, Hirji KF, Gekelman L, Jacobs AD, Champlin R . Efficacy of intensive chemotherapy for acute myelogenous leukemia associated with a preleukemic syndrome. J Clin Oncol 1989; 7: 1637–1645.
Legrand O, Perrot JY, Baudard M, Cordier A, Lautier R, Simonin G, Zittoun R, Casadevall N, Marie JP . The immunophenotype of 177 adults with acute myeloid leukemia: proposal of a prognostic score. Blood 2000; 96: 870–877.
Tanimoto M, Scheinberg DA, Cordon-Cardo C, Huie D, Clarkson BD, Old LJ . Restricted expression of an early myeloid and monocytic cell surface antigen defined by monoclonal antibody M195. Leukemia 1989; 3: 339–348.
Scheinberg DA, Tanimoto M, McKenzie S, Strife A, Old LJ, Clarkson BD . Monoclonal antibody M195: a diagnostic marker for acute myelogenous leukemia. Leukemia 1989; 3: 440–445.
McGraw KJ, Rosenblum MG, Cheung L, Scheinberg DA . Characterization of murine and humanized anti-CD33, gelonin immunotoxins reactive against myeloid leukemias. Cancer Immunol Immunother 1994; 39: 367–374.
Scheinberg DA, Lovett D, Divgi CR, Graham MC, Berman E, Pentlow K, Feirt N, Finn RD, Clarkson BD, Gee TS . A phase I trial of monoclonal antibody M195 in acute myelogenous leukemia: specific bone marrow targeting and internalization of radionuclide. J Clin Oncol 1991; 9: 478–490.
Schwartz MA, Lovett DR, Redner A, Finn RD, Graham MC, Divgi CR, Dantis L, Gee TS, Andreeff M, Old LJ . Dose-escalation trial of M195 labeled with iodine 131 for cytoreduction and marrow ablation in relapsed or refractory myeloid leukemias. J Clin Oncol 1993; 11: 294–303.
Caron PC, Co MS, Bull MK, Avdalovic NM, Queen C, Scheinberg DA . Biological and immunological features of humanized M195 (anti-CD33) monoclonal antibodies. Cancer Res 1992; 52: 6761–6767.
Caron PC, Jurcic JG, Scott AM, Finn RD, Divgi CR, Graham MC, Jureidini IM, Sgouros G, Tyson D, Old LJ . A phase 1B trial of humanized monoclonal antibody M195 (anti-CD33) in myeloid leukemia: specific targeting without immunogenicity. Blood 1994; 83: 1760–1768.
Xu Y, Scheinberg DA . Elimination of human leukemia by monoclonal antibodies in an athymic nude mouse leukemia model. Clin Cancer Res 1995; 1: 1179–1187.
Caron PC, Dumont L, Scheinberg DA . Supersaturating infusional humanized anti-CD33 monoclonal antibody HuM195 in myelogenous leukemia. Clin Cancer Res 1998; 4: 1421–1428.
Willman CL . The prognostic significance of the expression and function of multidrug resistance transporter proteins in acute myeloid leukemia: studies of the Southwest Oncology Group Leukemia Research Program. Semin Hematol 1997; 34: 25–33.
Maung ZT, MacLean FR, Reid MM, Pearson AD, Proctor SJ, Hamilton PJ, Hall AG . The relationship between bcl-2 expression and response to chemotherapy in acute leukaemia. Br J Haematol 1994; 88: 105–109.
Caron PC, Lai LT, Scheinberg DA . Interleukin-2 enhancement of cytotoxicity by humanized monoclonal antibody M195 (anti-CD33) in myelogenous leukemia. Clin Cancer Res 1995; 1: 63–70.
Grillo-Lopez AJ, White CA, Varns C, Shen D, Wei A, McClure A, Dallaire BK . Overview of the clinical development of rituximab: first monoclonal antibody approved for the treatment of lymphoma. Semin Oncol 1999; 26: 66–73.
Gianni L . Tolerability in patients receiving trastuzumab with or without chemotherapy. Ann Oncol 2001; 12 (Suppl. 1): S63–S68.
Jurcic JG, DeBlasio T, Dumont L, Yao TJ, Scheinberg DA . Molecular remission induction with retinoic acid and anti-CD33 monoclonal antibody HuM195 in acute promyelocytic leukemia. Clin Cancer Res 2000; 6: 372–380.
Vitale C, Romagnani C, Puccetti A, Olive D, Costello R, Chiossone L, Pitto A, Bacigalupo A, Moretta L, Mingari MC . Surface expression and function of p75/AIRM-1 or CD33 in acute myeloid leukemias: engagement of CD33 induces apoptosis of leukemic cells. Proc Natl Acad Sci USA 2001; 98: 5764–5769.
Mingari MC, Vitale C, Romagnani C, Falco M, Moretta L . p75/AIRM1 and CD33, two sialoadhesin receptors that regulate the proliferation or the survival of normal and leukemic myeloid cells. Immunol Rev 2001; 181: 260–268.
Sievers EL, Larson RA, Stadtmauer EA, Estey E, Lowenberg B, Dombret H, Karanes C, Theobald M, Bennett JM, Sherman ML, Berger MS, Eten CB, Loken MR, van Dongen JJ, Bernstein ID, Appelbaum FR . Efficacy and safety of gemtuzumab ozogamicin in patients with CD33-positive acute myeloid leukemia in first relapse. J Clin Oncol 2001; 19: 3244–3254.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Feldman, E., Kalaycio, M., Weiner, G. et al. Treatment of relapsed or refractory acute myeloid leukemia with humanized anti-CD33 monoclonal antibody HuM195. Leukemia 17, 314–318 (2003). https://doi.org/10.1038/sj.leu.2402803
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402803
Keywords
This article is cited by
-
Natural compounds targeting nuclear receptors for effective cancer therapy
Cancer and Metastasis Reviews (2023)
-
The progress and current status of immunotherapy in acute myeloid leukemia
Annals of Hematology (2017)
-
Driving CAR T-cells forward
Nature Reviews Clinical Oncology (2016)
-
Antibody-Based Treatment of Acute Myeloid Leukemia
Current Hematologic Malignancy Reports (2016)
-
Relapsed and Refractory Pediatric Acute Myeloid Leukemia: Current and Emerging Treatments
Pediatric Drugs (2014)