Abstract
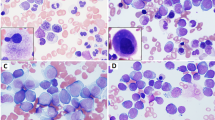
De novo acute myeloid leukemia (AML) with dysplastic features in erythroblasts, granulocytes and megakaryocytes, similar to those in myelodysplastic syndrome (MDS) has been described as AML with trilineage dysplasia (AML-TLD) since 1987. Several reports have suggested that AML-TLD is a subtype of de novo AML in adults and has a poor clinical outcome when treated by conventional chemotherapy. It is not certain whether allogeneic bone marrow transplantation (BMT) brings a favorable outcome for AML-TLD. To evaluate the therapeutic efficacy of allogeneic BMT for AML-TLD, we investigated the clinical data and outcomes of conventional chemotherapy and allogeneic BMT for 118 patients with de novo AML. These patients were registered consecutively for the Japan Adult Leukemia Study Group (JALSG) protocols at our institutes. We treated 28 AML-TLD patients and 90 AML-nonTLD patients with conventional chemotherapeutic protocols. AML-TLD patients did not have a significantly different complete remission (CR) rate (75.0% and 88.4% P = 0.1234), but had a significantly higher relapse rate than AML-nonTLD patients (94.1% and 49.3%, P = 0.0007). The outcome of chemotherapy for AML-TLD was significantly worse than that for AML-nonTLD. The overall survival (OS) and leukemia-free survival (LFS) at 6 years were 9.4% and 0% in AML-TLD group, and 51.9% (P = 0.0017) and 46.3% (P < 0.0001) in AML-nonTLD group, respectively. Meanwhile, among the patients who underwent allogeneic BMT, five of eight AML-TLD patients and eight of 14 AML-nonTLD patients were alive, and three and five patients survived more than 3 years, respectively. These results suggest that allogeneic BMT can improve the outcome for AML-TLD, which is poor when conventional chemotherapy is given alone. Allogeneic BMT before relapse may be the best therapeutic strategy for AML-TLD patients under 50 years of age if a donor is available.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Brito-Babapulle F, Catovsky D, Galton DA . Clinical and laboratory features of de novo acute myeloid leukaemia with trilineage myelodysplasia Br J Haematol 1987 66: 445–450
Jinnai I, Tomonaga M, Kuriyama K, Matsuo T, Nonaka H, Amenomori T, Yoshida Y, Kusano M, Tagawa M, Ichimaru M . Dysmegakaryocytopoiesis in acute leukaemias: its predominance in myelomonocytic (M4) leukaemia and implication for poor response to chemotherapy Br J Haematol 1987 66: 467–472
Estienne MH, Fenaux P, Preudhomme C, Lai JL, Zandecki M, Lepelley P, Cosson A . Prognostic value of dysmyelopoietic features in de novo acute myeloid leukemia: a report on 132 patients Clin Lab Haematol 1990 12: 57–65
Goasguen JE, Matsuo T, Cox C, Bennett JM . Evaluation of the dysmyelopoiesis in 336 patients with de novo acute myeloid leukaemia: major importance of dysgranulopoiesis for remission and survival Leukemia 1992 6: 520–525
Kuriyama K, Tomonaga M, Matsuo T, Kobayashi T, Miwa H, Shirakawa S, Tanimoto M, Adachi K, Emi N, Hiraoka A, Tominaga N, Imai K, Asou N, Tsubaki K, Takahashi I, Minami S, Yoshida M, Murakami H, Minato K, Oshima T, Furusawa S, Ohno R . Poor response to intensive chemotherapy in de novo acute myeloid leukaemia with trilineage myelodysplasia. Japan Adult Leukaemia Study Group (JALSG) Br J Haematol 1994 86: 767–773
Fukushima T, Kuriyama K, Yoshida S, Nagai K, Miyazaki Y, Moriuchi Y, Tomonaga M . Allogeneic bone marrow transplantation for two patients with acute myeloid leukemia with trilineage myelodysplasia (T-MDS) Rinsho Ketsueki 1994 35: 171–176
Ohno R, Kobayashi T, Tanimoto M, Hiraoka A, Imai K, Asou N, Tomonaga M, Tsubaki K, Takahashi I, Kodera Y, Yoshida M, Murakami H, Naoe T, Shimoyama M, Tsukada T, Takeo T, Teshima H, Onozawa Y, Fujimoto K, Kuriyama K, Horiuchi A, Kimura I, Minami S, Miura Y, Kageyama S, Tahara T, Masaoka T, Shirakawa S, Saito H . Randomized study of individualized induction therapy with or without vincristine, and of maintenance-intensification therapy between 4 or 12 courses in adult acute myelid leukemia Cancer 1993 71: 3888–3895
Kobayashi T, Miyawaki S, Tanimoto M, Kuriyama K, Murakami H, Yoshida M, Minami S, Minato K, Tsubaki K, Ohmoto E, Oh H, Jinnai I, Sakamaki H, Hiraoka A, Kanamaru A, Takahashi I, Saito K, Naoe T, Yamada O, Asou N, Kageyama S, Emi N, Matsuoka A, Tomonaga M, Saito H, Ueda R, Ohno R . Randomized trials between behenoyl cytarabine and cytarabine in combination induction and consolidation therapy, and with or without ubenimex after maintenance/intensification therapy in adult acute myeloid leukemia. The Japan Leukemia Study Group J Clin Oncol 1996 14: 204–213
Ohno R, Miyawaki S, Hatake K, Kuriyama K, Saito K, Kanamaru A, Kobayashi T, Kodera Y, Nishikawa K, Matsuda S, Yamada O, Omoto E, Takeyama H, Tsukuda K, Asou N, Tanimoto M, Shiozaki H, Tomonaga M, Masaoka T, Miura Y, Takaku F, Ohashi Y, Motoyoshi K . Human urinary macrophage colony-stimulating factor reduces the incidence and duration of febrile neutropenia and shortens the period required to finish three courses of intensive consolidation therapy in acute myeloid leukemia: a double-blind controlled study J Clin Oncol 1997 15: 2954–2965
Tutschka PJ, Copelan EA, Klein JP . Bone marrow transplantation for leukemia following a new busulfan and cyclophosphamide regimen Blood 1987 70: 1382–1388
Appelbaum FR, Barrall J, Storb R, Fisher LD, Schoch G, Ramberg RE, Shulman H, Anasetti C, Bearman SI, Beatty P, Bensinger WI, Buckner CD, Clift RA, Hansen JA, Martin P, Petersen FB, Sanders JE, Singer J, Stewart P, Sullivan KM, Witherspoon RP, Thomas ED . Bone marrow transplantation for patients with myelodysplasia. Pretreatment variables and outcome Ann Intern Med 1990 112: 590–597
Storb R, Deeg HJ, Whitehead J, Appelbaum F, Beatty P, Bensinger W, Buckner CD, Clift R, Doney K, Farewell V, Hansen J, Hill R, Lum L, Martin P, McGuffin R, Sanders J, Stewart P, Sullivan K, Witherspoon R, Yee G, Thomas D . Methotrexate and cyclosporine compared with cyclosporine alone for prophylaxis of acute graft-versus-host disease after marrow transplantation for leukemia New Engl J Med 1986 314: 729–735
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA, Lerner KG, Thomas ED . Clinical manifestations of graft-versus-host disease in human recipients from HLA-matched sibling donors Transplantation 1974 18: 295–304
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR, Sultan C . Proposals for the classification of the acute leukaemias Br J Haematol 1976 33: 451–458
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR, Sultan C . Proposed revised criteria for the classification of acute myeloid leukemia. A report of the French–American–British Cooperative Group Ann Intern Med 1985 103: 620–625
Grimwade D, Walker H, Oliver F, Wheatley K, Harrison C, Harrison G, Rees J, Hann I, Stevens R, Burnett A, Goldstone A . The importance of diagnostic cytogenetics on outcome in AML: analysis of 1612 patients entered into the MRC AML 10 trial. The Medical Research Council Adult and Children's Leukemia Working Parties Blood 1998 92: 2322–2333
Buchner T, Hiddemann W, Wormann B, Loffler H, Gassmann W, Haferlach T, Fonatsch C, Haase D, Schoch C, Hossfeld D, Lengfelder E, Aul C, Heyll A, Maschmeyer G, Ludwig WD, Sauerland MC, Heinecke A . Double induction strategy for acute myeloid leukemia: the effect of high-dose cytarabine with mitoxantrone instead of standard-dose cytarabine with daunorubicin and 6-thioguanine: a randomized trial by the German AML Cooperative Group Blood 1999 93: 4116–4124
Kuriyama K, Miyazaki Y, Arimura K, Nagai K, Nakamura H, Matsuo T, Tomonaga M . Morphological comparison of dysplastic changes between de novo acute myeloid leukemia (AML) with trilineage myelodysplasia and AML developed from de novo myelodysplastic syndromes Leuk Res 1995 19: 121–125
Gassmann W, Schmitz N, Loffler H, De Witte T . Intensive chemotherapy and bone marrow transplantation for myelodysplastic syndromes Semin Hematol 1996 33: 196–205
Armitage JO, Dick FR, Needleman SW, Burns CP . Effect of chemotherapy for the dysmyelopoietic syndrome Cancer Treat Rev 1981 65: 601–605
De Witte T, Muus P, De Pauw B, Haanen C . Intensive antileukemic treatment of patients younger than 65 years with myelodysplastic syndromes and secondary acute myelogenous leukemia Cancer 1990 66: 831–837
Fenaux P, Morel P, Rose C, Lai JL, Jouet JP, Bauters F . Prognostic factors in adult de novo myelodysplastic syndromes treated by intensive chemotherapy Br J Haematol 1991 77: 497–501
Longmore G, Guinan EC, Weinstein HJ, Gelber RD, Rappeport JM, Antin JH . Bone marrow transplantation for myelodysplasia and secondary acute nonlymphoblastic leukemia J Clin Oncol 1990 8: 1707–1714
De Witte T, Gratwohl A . Bone marrow transplantation for myelodysplastic syndrome and secondary leukaemias Br J Haematol 1993 84: 361–364
De Witte T, Zwaan F, Hermans J, Vernant J, Kolb H, Vossen J, Lonnqvist B, Beelen D, Ferrant A, Gmur J, Liu Yin J, Troussard X, Cahn J, Van Lint M, Gratwohl A . Allogeneic bone marrow transplantation for secondary leukaemia and myelodysplastic syndrome: a survey by the Leukaemia Working Party of the European Bone Marrow Transplantation Group (EBMTG) Br J Haematol 1990 74: 151–155
Anderson JE, Gooley TA, Schoch G, Anasetti C, Bensinger WI, Clift RA, Hansen JA, Sanders JE, Storb R, and Applebaum FR . Stem cell transplantation for secondary acute myeloid leukemia: evaluation of transplantation as initial therapy or following induction chemotherapy Blood 1997 89: 2578–2585
Acknowledgements
The following institutes participated in this study: Department of Hematology, Nagasaki University Hospital, Nagasaki, Sasebo City General Hospital, Sasebo, Nagasaki-Chuo National Hospital, Ohmura, Nagasaki Municipal Medical Center, Nagasaki and St Francis Hospital, Nagasaki. Allogeneic BMT was performed in Nagasaki University and Sasebo City General Hospital. We are grateful to the JALSG statistical office for providing a portion of the data on AML patients.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Taguchi, J., Miyazaki, Y., Yoshida, S. et al. Allogeneic bone marrow transplantation improves the outcome of de novo AML with trilineage dysplasia (AML-TLD). Leukemia 14, 1861–1866 (2000). https://doi.org/10.1038/sj.leu.2401924
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2401924
Keywords
This article is cited by
-
Morphological diagnoses of the Japan Adult Leukemia Study Group acute myeloid leukemia protocols: Central review
International Journal of Hematology (2001)