Abstract
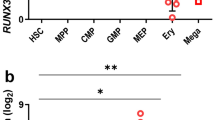
The FLI-1 oncogene, a member of the ETS family of transcription factors, is associated with both normal and abnormal hematopoietic cell growth and lineage-specific differentiation. We have previously shown that overexpression of FLI-1 in pluripotent human hematopoietic cells leads to the induction of a megakaryocytic phenotype. In this report we show that FLI-1 also acts as an inhibitor of erythroid differentiation. Following the induction of erythroid differentiation, pluripotent cells express reduced levels of FLI-1. In contrast, when FLI-1 is overexpressed in these cells, the levels of erythroid markers are reduced. The ability of FLI-1 overexpressing cells to respond to erythroid-specific inducers such as hemin and Ara-C is also inhibited, and the uninduced cells show a reduced level of the erythroid-associated GATA-1 transcription factor mRNA. Furthermore, expression of a GATA-1 promoter-driven reporter construct in K562 cells is inhibited by co-transfection with a construct expressing FLI-1. Our results support the hypothesis that FLI-1 can act both positively and negatively in the regulation of hematopoietic cell differentiation, and that inhibition of GATA-1 expression may contribute to FLI-1-mediated inhibition of erythroid differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Quang CT, Pironin M, von Lindern M, Beug H, Ghysdael J . Spi-1 and mutant p53 regulate different aspects of the proliferation and differentiation control of primary erythroid progenitors Oncogene 1995 11: 1229–1239
Carrere S, Verger A, Flourens A, Stehelin D, Duterque-Coquillaud M . Erg proteins, transcription factors of the Ets family, form homo, heterodimers and ternary complexes via two distinct domains Oncogene 1998 16: 3261–3268
Anderson MK, Hernandez-Hoyos G, Diamond RA, Rothenberg EV . Precise developmental regulation of Ets family transcription factors during specification and commitment to the T cell lineage Development 1999 126: 3131–3148
Nunn M, Weiher H, Bullock P, Duesberg P . Avian erythroblastosis virus E26: nucleotide sequence of the tripartite onc gene and of the LTR, and analysis of the cellular prototype of the viral ets sequence Virology 1984 139: 330–339
Moscovici C, Samarut J, Gazzolo L, Moscovici MG . Myeloid and erythroid neoplastic responses to avian defective leukemia viruses in chickens and in quail Virology 1981 113: 765–768
Yuan CC, Kan N, Dunn KJ, Papas TS, Blair DG . Properties of a murine retroviral recombinant of avian acute leukemia virus E26: a murine fibroblast assay for v-ets function J Virol 1989 63: 205–215
Ben-David Y, Bernstein A . Friend virus-induced erythroleukemia and the multistage nature of cancer Cell 1991 66: 831–834
Ben-David Y, Giddens EB, Letwin K, Bernstein A . Erythroleukemia induction by Friend murine leukemia virus: insertional activation of a new member of the ets gene family, Fli-1, closely linked to c-ets-1 Genes Dev 1991 5: 908–918
Ott DE, Keller J, Rein A . 10A1 MuLV induces a murine leukemia that expresses hematopoietic stem cell markers by a mechanism that includes fli-1 integration Virology 1994 205: 563–568
Delattre O, Zucman J, Plougastel B, Desmaze C, Melot T, Peter M, Kovar H, Joubert I, de Jong P, Rouleau G et al. Gene fusion with an ETS DNA-binding domain caused by chromosome translocation in human tumours Nature 1992 359: 162–165
Dittmer J, Nordheim A . Ets transcription factors and human disease Biochim Biophys Acta 1998 1377: F1–11
Starck J, Doubeikovski A, Sarrazin S, Gonnet C, Rao G, Skoultchi A, Godet J, Dusanter-Fourt I, Morle F . Spi-1/PU.1 is a positive regulator of the Fli-1 gene involved in inhibition of erythroid differentiation in friend erythroleukemic cell lines Mol Cell Biol 1999 19: 121–135
Klemsz MJ, McKercher SR, Celada A, Van Beveren C, Maki RA . The macrophage and B cell-specific transcription factor PU.1 is related to the ets oncogene Cell 1990 61: 113–124
Moreau-Gachelin F, Tavitian A, Tambourin P . Spi-1 is a putative oncogene in virally induced murine erythroleukaemias Nature 1988 331: 277–280
Watson DK, Smyth FE, Thompson DM, Cheng JQ, Testa JR, Papas TS, Seth A . The ERGB/Fli-1 gene: isolation and characterization of a new member of the family of human ETS transcription factors Cell Growth Differ 1992 3: 705–713
Athanasiou M, Clausen PA, Mavrothalassitis GJ, Zhang XK, Watson DK, Blair DG . Increased expression of the ETS-related transcription factor FLI-1/ERGB correlates with and can induce the megakaryocytic phenotype Cell Growth Differ 1996 7: 1525–1534
Shivdasani RA, Fujiwara Y, McDevitt MA, Orkin SH . A lineage-selective knockout establishes the critical role of transcription factor GATA-1 in megakaryocyte growth and platelet development EMBO J 1997 16: 3965–3973
McKercher SR, Torbett BE, Anderson KL, Henkel GW, Vestal DJ, Baribault H, Klemsz M, Feeney AJ, Wu GE, Paige CJ, Maki RA . Targeted disruption of the PU.1 gene results in multiple hematopoietic abnormalities EMBO J 1996 15: 5647–5658
Takahashi S, Komeno T, Suwabe N, Yoh K, Nakajima O, Nishimura S, Kuroha T, Nagasawa T, Yamamoto M . Role of GATA-1 in proliferation and differentiation of definitive erythroid and megakaryocytic cells in vivo Blood 1998 92: 434–442
Tsang AP, Fujiwara Y, Hom DB, Orkin SH . Failure of megakaryopoiesis and arrested erythropoiesis in mice lacking the GATA-1 transcriptional cofactor FOG Genes Dev 1998 12: 1176–1188
Valtieri M, Tocci A, Gabbianelli M, Luchetti L, Masella B, Vitelli L, Botta R, Testa U, Condorelli GL, Peschle C . Enforced TAL-1 expression stimulates primitive, erythroid and megakaryocytic progenitors but blocks the granulopoietic differentiation program Cancer Res 1998 58: 562–569
Rowley PT, Ohlsson-Wilhelm BM, Farley BA, LaBella S . Inducers of erythroid differentiation in K562 human leukemia cells Exp Hematol 1981 9: 32–37
Athanasiou M, Mavrothalassitis GJ, Yuan CC, Blair DG . The Gag-Myb-Ets fusion oncogene alters the apoptotic response and growth factor dependence of interleukin-3 dependent murine cells Oncogene 1996 12: 337–344
Martin P, Papayannopoulou T . HEL cells: a new human erythroleukemia cell line with spontaneous and induced globin expression Science 1982 216: 1233–1235
Andersson LC, Jokinen M, Gahmberg CG . Induction of erythroid differentiation in the human leukaemia cell line K562 Nature 1979 278: 364–365
Romano-Spica V, Suzuki H, Georgiou P, Chen S-L, Ascione R, Papas TS, Bhat NK . Expression of ets family genes in hematopoietic cells Intl J Oncol 1994 4: 521–531
Jarvinen M . Vimentin in human erythroleukemia (HEL) cells is modulated with differentiation inducers Cell Biol Int Rep 1990 14: 199–209
Clausen P, Athanasiou M, Chen Z-Q, Dunn KJ, Zhang Q, Lautenberger JA, Mavrothalassitis G, Blair DG . ETS-1 induces increased expression of erythroid markers in the pluripotent erythroleukemic cell lines K562 and HEL Leukemia 1997 11: 1224–1233
Andersson LC, Jokinen M, Klein E, Klein G, Nilsson K . Presence of erythrocytic components in the K562 cell line (letter) Int J Cancer 1979 24: 514
Gahmberg CG, Jokinen M, Andersson LC . Expression of the major red cell sialoglycoprotein, glycophorin A, in the human leukemic cell line K562 J Biol Chem 1979 254: 7442–7448
Rutherford TR, Clegg JB, Weatherall DJ . K562 human leukaemic cells synthesise embryonic haemoglobin in response to haemin Nature 1979 280: 164–165
Pevny L, Lin CS, D'Agati V, Simon MC, Orkin SH, Costantini F . Development of hematopoietic cells lacking transcription factor GATA-1 Development 1995 121: 163–172
Weiss MJ, Orkin SH . GATA transcription factors: key regulators of hematopoiesis Exp Hematol 1995 23: 99–107
Orkin SH . Hematopoiesis: how does it happen? Curr Opin Cell Biol 1995 7: 870–877
Sun-Hoffman L, Aurigemma RE, Sun B, Ruscetti SK . Transactivation of the GATA-1 promoter by a myb-ets-containing mouse retrovirus is mediated by CACCC elements Oncogene 1996 13: 1037–1042
Baron MH, Farrington SM . Positive regulators of the lineage-specific transcription factor GATA-1 in differentiating erythroid cells Mol Cell Biol 1994 14: 3108–3114
Tsai SF, Strauss E, Orkin SH . Functional analysis and in vivo footprinting implicate the erythroid transcription factor GATA-1 as a positive regulator of its own promoter Genes Dev 1991 5: 919–931
Robert-Lezenes J, Meneceur P, Ray D, Moreau-Gachelin F . Protooncogene expression in normal, preleukemic, and leukemic murine erythroid cells and its relationship to differentiation and proliferation (published erratum appears in Cancer Res 1988 Sep 1; 48(17): 5056) Cancer Res 1988 48: 3972–3976
Ben-David Y, Giddens EB, Bernstein A . Identification and mapping of a common proviral integration site Fli-1 in erythroleukemia cells induced by Friend murine leukemia virus Proc Natl Acad Sci USA 1990 87: 1332–1336
Kwiatkowski BA, Bastian LS, Bauer TR Jr, Tsai S, Zielinska-Kwiatkowska AG, Hickstein DD . The ets family member Tel binds to the Fli-1 oncoprotein and inhibits its transcriptional activity J Biol Chem 1998 273: 17525–17530
Rekhtman N, Radparvar F, Evans T, Skoultchi AI . Direct interaction of hematopoietic transcription factors PU.1 and GATA- 1: functional antagonism in erythroid cells Genes Dev 1999 13: 1398–1411
Pereira R, Quang CT, Lesault I, Dolznig H, Beug H, Ghysdael J . FLI-1 inhibits differentiation and induces proliferation of primary erythroblasts Oncogene 1999 18: 1597–1608
Tamir A, Howard J, Higgins RR, Li YJ, Berger L, Zacksenhaus E, Reis M, Ben-David Y . Fli-1, an ets-related transcription factor, regulates erythropoietin-induced erythroid proliferation and differentiation: evidence for direct transcriptional repression of the Rb gene during differentiation Mol Cell Biol 1999 19: 4452–4464
Plumb M, Frampton J, Wainwright H, Walker M, Macleod K, Goodwin G, Harrison P . GATAAG; a cis-control region binding an erythroid-specific nuclear factor with a role in globin and non-globin gene expression Nucleic Acids Res 1989 17: 73–92
Romeo PH, Prandini MH, Joulin V, Mignotte V, Prenant M, Vainchenker W, Marguerie G, Uzan G . Megakaryocytic and erythrocytic lineages share specific transcription factors Nature 1990 344: 447–449
Orkin SH . GATA-binding transcription factors in hematopoietic cells Blood 1992 80: 575–581
Pevny L, Simon MC, Robertson E, Klein WH, Tsai SF, D'Agati V, Orkin SH, Costantini F . Erythroid differentiation in chimaeric mice blocked by a targeted mutation in the gene for transcription factor GATA-1 Nature 1991 349: 257–260
McNagny KM, Sieweke MH, Doderlein G, Graf T, Nerlov C . Regulation of eosinophil-specific gene expression by a C/EBP-Ets complex and GATA-1 EMBO J 1998 17: 3669–3680
Minami T, Tachibana K, Imanishi T, Doi T . Both Ets-1 and GATA-1 are essential for positive regulation of platelet factor 4 gene expression Eur J Biochem 1998 258: 879–889
Moreau-Gachelin F, Ray D, Mattei MG, Tambourin P, Tavitian A . The putative oncogene Spi-1: murine chromosomal localization and transcriptional activation in murine acute erythroleukemias (published erratum appears in Oncogene 1990 Jun; 5(6): 941) Oncogene 1989 4: 1449–1456
Delgado MD, Gutierrez P, Richard C, Cuadrado MA, Moreau-Gachelin F, Leon J . Spi-1/PU.1 proto-oncogene induces opposite effects on monocytic and erythroid differentiation of K562 cells Biochem Biophys Res Commun 1998 252: 383–391
Yamada T, Kondoh N, Matsumoto M, Yoshida M, Maekawa A, Oikawa T . Overexpression of PU.1 induces growth and differentiation inhibition and apoptotic cell death in murine erythroleukemia cells Blood 1997 89: 1383–1393
Acknowledgements
We wish to thank Karen Cannon for preparation of the manuscript, Louise Finch, Flow Cytometry Laboratory, Clinical Services Program, SAIC, for performing flow cytometry analysis, and Sandra Ruscetti for providing GATA-1 promoter constructs and for helpful discussions. This project has been funded in whole or in part with Federal funds from the National Cancer Institute, National Institutes of Health, under Contract No. N01-CO-56000. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the US Government.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Athanasiou, M., Mavrothalassitis, G., Sun-Hoffman, L. et al. FLI-1 is a suppressor of erythroid differentiation in human hematopoietic cells. Leukemia 14, 439–445 (2000). https://doi.org/10.1038/sj.leu.2401689
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2401689
Keywords
This article is cited by
-
In vitro erythrocyte production using human-induced pluripotent stem cells: determining the best hematopoietic stem cell sources
Stem Cell Research & Therapy (2023)
-
ETV6 dependency in Ewing sarcoma by antagonism of EWS-FLI1-mediated enhancer activation
Nature Cell Biology (2023)
-
Current insights into the role of Fli-1 in hematopoiesis and malignant transformation
Cellular and Molecular Life Sciences (2022)
-
Identification of diterpenoid compounds that interfere with Fli-1 DNA binding to suppress leukemogenesis
Cell Death & Disease (2019)
-
GWAS of mosaic loss of chromosome Y highlights genetic effects on blood cell differentiation
Nature Communications (2019)