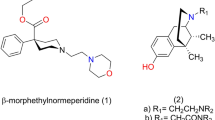
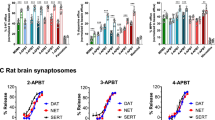
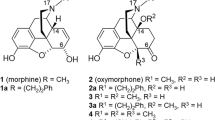
Abstract
WE wish to describe the preliminary pharmacology of a mescaline analogue, 2-(3,4,5-trimethoxyphenyl)-cyclopropylamine (TMT, II). This compound was selected for synthesis as a potential long-acting psychotomimetic which would be useful for studying the mode of action of mescaline-like compounds.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Shulgin, A. T., Bunnell, S., and Sargent, T., Nature, 189, 1011 (1961).
Kaiser, C., Trost, B. M., Beeson, J., and Weinstock, J., J. Org. Chem., 30, 3972 (1965).
Weinstock, J., J. Org. Chem., 26, 3511 (1961).
Melander, B., and Gliniecke, G., Acta Pharmacol. Toxicol., 18, 239 (1961).
Noteboom, L., Proc. Roy. Acad., Amsterdam, 37, 562 (1934).
Michaux, R., and Verly, W. G., Life Sci., 3, 175 (1963).
Ernst, A. M., Psychopharmacol., 7, 381 (1965).
Schiele, B. C., Psychosomatic Medicine, First Hahneman Symposium (Lea and Febiger, Philadelphia, 1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WALTERS, G., COOPER, P. Alicyclic Analogue of Mescaline. Nature 218, 298–300 (1968). https://doi.org/10.1038/218298a0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1038/218298a0
This article is cited by
-
Stereochemical Requirements of the Mescaline Receptor
Nature (1972)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.