Abstract
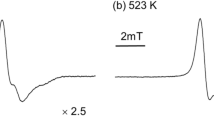
WE present here the results of an electron spin resonance investigation of the free radicals formed from diethyl-hydroxylamine, dioctylhydroxylamine and N-hydroxypyrrolidine. Dioctylhydroxylamine was prepared by the method of Harris and Olcott1. Diethylhydroxylamine was obtained from Pennsalt Chemicals (85 per cent and 100 per cent) and from K and K Laboratories (85 per cent). N-Hydroxypyrrolidine was prepared by the method Wolfenstein2 used for N-hydroxypiperidne and by the method described by Blout3.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Harris, L. A., and Olcott, H. S., J. Amer. Oil Chem. Soc., 43, 11 (1966).
Wolfenstein, R., Chem. Ber., 25, 2777 (1892).
Blout, E. R., Cohen, S. G., and Green, M., US Patent 2,843,481, July 15, 1958.
Coppinger, G. M., and Swalen, J. D., J. Amer. Chem. Soc., 83, 4900 (1961).
Chapelet-Letourneurx, G., Lemaire, H., and Rassat, A., Bull. Soc. Chim., 3283 (1965).
Adams, J. Q., Nicksic, S. W., and Thomas, J. R., J. Chem. Phys., 45, 654 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WEIL, J., WINDLE, J. Electron Spin Resonance Study of Nitroxide Radicals from Diethylhydroxylamine, Dioctylhydroxylamine and N-Hydroxypyrrolidine. Nature 217, 842–843 (1968). https://doi.org/10.1038/217842b0
Received:
Issue Date:
DOI: https://doi.org/10.1038/217842b0
This article is cited by
-
Aliphatic hydroxylamines as lipid antioxidants
Lipids (1970)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.