Abstract
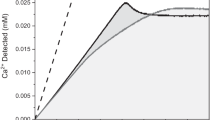
THE formation of white spots, which is the first clinical sign of dental caries, is a consequence of sub-surface decalcification1. The carious process therefore involves dissolution of the crystallites—predominantly hydroxyapatite—which constitute the bulk (about 95 per cent by weight) of human dental enamel2. Knowledge of the dissolution kinetics of hydroxyapatite is thus a prerequisite for a complete understanding of the aetiology of dental caries. Moreover, a study of the influence of certain cariostatic agents, such as organic phosphate salts3,4, on the dissolution of hydroxyapatite could well clarify their possible mode(s) of action because those agents which retard dissolution could well possess prophylactic cariostatic activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Darling, A. J., Brit. Dental J., 101, 289 (1956).
Carlström, D., Adv. Oral Biol., 1, 259 (1964).
Gagolski, J., and Lilienthal, B., Austral. Patent No. 256211 (1965; filed July, 1961).
McClure, F. C., Science, 144, 1337 (1964).
Perloff, A., and Posner, A. S., Science, 124, 583 (1956).
Rootare, H. M., Deitz, V. R., and Carpenter, F. G., J. Colloid Sci., 17, 179 (1962).
Kanevskii, E. A., and Pchelkin, V. A., Kinetika i Kataliz, 2, 188 (1964).
McCann, H. G., J. Biol. Chem., 201, 247 (1953).
Middleton, K. R., Analyst, 86, 111 (1961).
Gee, A., Domingues, L. P., and Deitz, V. R., Anal. Chem., 26, 1487 (1954).
Brønsted, J. N., Z. Physik. Chem., 103, 307 (1922).
Frank, F. C., Disc. Farad. Soc., 5, 68 (1949).
Ives, M. B., and Hirth, J. P., J. Chem. Phys., 33, 517 (1960).
Miura, M., and Naono, H., Bull. Chem. Soc. Japan, 38, 492 (1965).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BRADY, B., NAPPER, D. & SMYTHE, B. Dissolution Kinetics of Hydroxyapatite. Nature 212, 77–78 (1966). https://doi.org/10.1038/212077a0
Issue Date:
DOI: https://doi.org/10.1038/212077a0
This article is cited by
-
Applications of phytic acid
Journal of the American Oil Chemists' Society (1983)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.