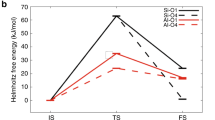
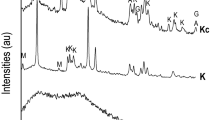
Abstract
CALCIUM aluminate hydrates have recently been reported to form as products of reaction between aluminium-bearing clay minerals and calcium hydroxide at or near room temperatures1,2. The investigation recorded here was initiated to determine whether hydrous alumina would react with calcium hydroxide under similar conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Glenn, G. R., and Handy, R. L., Highway Res. Bd. Rec., 29, 70 (1963).
Diamond, S., White, J. L., and Dolch, W. L., in Proc. Twelfth Nat. Clay Conf., edit. by Bradley, W. F. (Pergamon Press, New York, 1964; in the press).
Roberts, M. H., J. App. Chem., 543 (1957).
Seligmann, P., and Greening, N. R., J. Res. Develop. Lab., Portland Cement Assoc., 4, 2 (1962).
Alegre, R., Rev. Mater. Constr. Trav. Publics, 301 (1963).
Hunt, C. M., thesis, Univ. Maryland (1959).
Buttler, F. G., Dent-Glasser, L. S., and Taylor, H. F. W., J. Amer. Ceram. Soc., 42, 121 (1959).
Feitknecht, W., and Gerber, M., Helv. Chim. Acta, 25, 131 (1942).
Mortland, M. M., and Gastuche, M. C., C. R. Acad. Sci., Paris, 255, 2131 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DIAMOND, S. Rapid Reaction of Lime with Hydrous Alumina. Nature 204, 183–185 (1964). https://doi.org/10.1038/204183a0
Issue Date:
DOI: https://doi.org/10.1038/204183a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.