Abstract
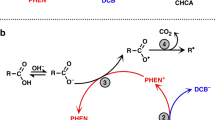
RED chemiluminescence corresponding to about 40 kcal/mol., and attributable to the forbidden transition 1Σ+g → 3Σ−g of the oxygen molecule, has been observed in three recent investigations1–3, but no explanation of the effect has been presented. These involve organic hydroperoxides or oxidations with hydrogenperoxide and also the reaction of hydrogen peroxide with HClO. In contrast with this, decompositions of hydrogen peroxide cataclysed by copper ions or by platinum show no trace of the effect, and in the photochemical decomposition of ozone ground state oxygen molecules are formed with vibrational energies up to 70 kcal/mol. (ref. 4). The state of newly formed oxygen molecules is evidently not determined solely by energy factors. Since in the thermal decomposition of an organic peroxide to a ketone (or quinine)  about 70 kcal/mol. is liberated it is very possible that the ketosis (K) is formed in its triplet state. The spin-allowed reaction 3K + 3O2 → 1K + 1O2 would then account for the formation of singlet excited oxygen molecule.
about 70 kcal/mol. is liberated it is very possible that the ketosis (K) is formed in its triplet state. The spin-allowed reaction 3K + 3O2 → 1K + 1O2 would then account for the formation of singlet excited oxygen molecule.
Similar content being viewed by others
Article PDF
References
Khan, A. u., and Kasha, M., J. Chem. Phys., 39, 2105 (1963).
Bowen, E. J., and Lloyd, R. A., Proc. Roy. Soc., A, 275, 465 (1963).
Bowen, E. J., and Lloyd, R. A., Proc. Chem. Soc., 305 (1963).
McGrath, W. D., and Norrish, R. G. W., Proc. Roy. Soc., A, 254, 317 (1960).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BOWEN, E. Chemiluminescence from Dissolved Oxygen. Nature 201, 180 (1964). https://doi.org/10.1038/201180b0
Issue Date:
DOI: https://doi.org/10.1038/201180b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.