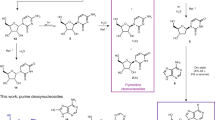
Abstract
THE release of purines and characteristic sequences of pyrimidine oligonucleotides from DNA has been achieved by the use of diphenylamine and formic acid1. In RNA the phosphodiester bonds are extremely acid and alkali-sensitive due to the ease with which cyclic phosphate intermediates are formed involving the 2′ hydroxyl group. In order to limit the possibility of cyclization an attempt was made to carry out a depurination reaction with RNA in the solid phase.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Burton, K., and Petersen, G. B., Biochem. J., 75, 17 (1960).
Bramwell, M. E., and Jones, A. S., Nature, 195, 1095 (1962).
Crestfield, A. M., Smith, K. C., and Allen, F. W., J. Biol. Chem., 216, 185 (1955).
Levene, P. A., and Stiller, E. T., J. Biol. Chem., 104, 299 (1934).
Hershey, A. D., Dixon, J., and Chase, M., J. Gen. Physiol., 36, 777 (1953).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BRAMWELL, M. Reaction of Hydrogen Chloride Gas on RNA and its Component Nucleotides. Nature 199, 811–812 (1963). https://doi.org/10.1038/199811b0
Issue Date:
DOI: https://doi.org/10.1038/199811b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.