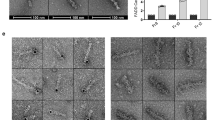
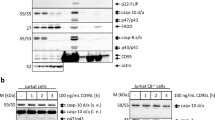
Abstract
Fas is a cell-surface receptor molecule that relays apoptotic (cell death) signals into cells. When Fas is activated by binding of its ligand, the proteolytic protein caspase-8 is recruited to a signalling complex known as DISC by binding to a Fas-associated adapter protein. A large new protein, FLASH, has now been identified by cloning of its complementary DNA. This protein contains a motif with oligomerizing activity whose sequence is similar to that of the Caenorhabditis elegans protein CED-4, and another domain (DRD domain) that interacts with a death-effector domain in caspase-8 or in the adapter protein. Stimulated Fas binds FLASH, so FLASH is probably a component of the DISC signalling complex. Transient expression of FLASH activates caspase-8, whereas overexpression of a truncated form of FLASH containing only one of its DRD or CED-4-like domains does not allow activation of caspase-8 and Fas-mediated apoptosis to occur. Overexpression of full-length FLASH blocks the anti-apoptotic effect of the adenovirus protein E1B19K. FLASH is therefore necessary for the activation of caspase-8 in Fas-mediated apoptosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Itoh, N.et al. The polypeptide encoded by the cDNA for human cell surface antigen Fas can mediate apoptosis. Cell 66, 233– 243 (1991).
Nagata, S. Apoptosis by death factor. Cell 88, 355– 365 (1997).
Yonehara, S., Ishii, A. & Yonehara, M. Acell-killing monoclonal antibody (anti-Fas) to a cell surface antigen co-downregulated with the receptor of tumor necrosis factor. J. Exp. Med. 169, 1747– 1756 (1989).
Trauth, B. C.et al. Monoclonal antibody-mediated tumor regression by induction of apoptosis. Science 245, 301– 305 (1989).
Suda, T., Takahashi, T., Golstein, P. & Nagata, S. Molecular cloning and expression of the Fas ligand, a novel member of the tumor necrosis factor family. Cell 75, 1169 –1178 (1993).
Chinnaiyan, A. M., O'Rourke, K., Tewari, M. & Dixit, V. M. FADD, a novel death domain-containing protein, interacts with the death domain of Fas and initiates apoptosis. Cell 81, 505– 512 (1995).
Boldin, M. P.et al. Anovel protein that interacts with the death domain of Fas/APO1 contain a sequence motif related to the death domain. J. Biol. Chem. 270, 7795–7798 ( 1995).
Yeh, W. C.et al. FADD: essential for embryo development and signaling from some, but not all, inducers of apoptosis. Science 279, 1954–1958 (1998).
Zhang, J.et al. Fas-mediated apoptosis and activation-induced T-cell proliferation are defective in mice lacking FADD/Mort1. Nature 392, 296–300 (1998).
Kischkel, F. C.et al . Cytotoxicity-dependent APO-1 (Fas/CD95)-associated proteins form a death-inducing signaling complex (DISC) with the receptor. EMBO J. 14, 5579–5588 ( 1995).
Boldin, M. P., Goncharov, T. M., Goltsev, Y. V. & Wallach, D. Involvement of MACH, a novel MORT1/FADD-interacting protease, in Fas/APO-1- and TNF receptor-induced cell death. Cell 85, 803–815 (1996).
Muzio, M.et al. FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death-inducing signaling complex. Cell 85, 817–827 ( 1996).
Muzio, M.et al. An induced proximity model for caspase-8 activation. J. Biol. Chem. 273, 2926–2930 (1998).
Yang, X., Chang, H. Y. & Baltimore, D. Autoproteolytic activation of pro-caspases by oligomerization. Mol. Cell 1, 319–325 (1998).
Enari, M., Talanian, R. V., Wong, W. W. & Nagata, S. Sequential activation of ICE-like and CPP32-like proteases during Fas-mediated apoptosis. Nature 380, 723– 726 (1996).
Takahashi, A.et al. Affinity labeling displays the stepwise activation of ICR-related proteases by Fas, staurosporine, and CrmA-sensitive caspase-8. Oncogene 14, 2741–2752 ( 1997).
Fernandes-Alnemri, T., Litwack, G. & Alnemri, E. S. Mch2, a new member of the apoptotic Ced-3/Ice cysteine protease gene family. Cancer Res. 55, 2737 –2742 (1995).
Villa, P., Kaufmann, S. H. & Earnshaw, W. C. Caspases and caspase inhibitors. Trends Biochem. Sci. 22, 388–393 (1997).
Rudel, T. & Bokoch, G. M. Membrane and morphological changes in apoptotic cells regulated by caspase-mediated activation of PAK2. Science 276, 1571–1571 ( 1997).
Enari, M.et al. Acaspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature 391, 43– 50 (1998).
Chinnaiyan, A. M.et al. Role of CED-4 in the activation of CED-3. Nature 388, 728–729 ( 1997).
Li, P.et al. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91, 479–489 (1997).
Perez, D. & White, E. E1B 19K inhibits Fas-mediated apoptosis through FADD-dependent sequestration of FLICE. J. Cell Biol. 141, 1255–1266 (1998).
Scaffidi, C.et al. Two CD95 (APO-1/Fas) signaling pathways. EMBO J. 17, 1675–1687 ( 1998).
Kozak, M. The scanning model for translation: an update. J. Cell Biol. 108, 229–241 (1989).
Walker, J. E., Saraste, M., Runswick, M. J. & Gay, N. J. Distantly related sequences in the alpha- and beta-subunits of ATP synthetase, myosin, kinases and other ATP-requiring enzymes and a common nucleotide binding fold. EMBO J. 1, 945–951 (1982).
Nigg, E. A. Nucleocytoplasmic transport: signals, mechanisms and regulation. Nature 386, 779–787 ( 1997).
Wada, A., Fukuda, M., Mishima, M. & Nishida, E. Nuclear export of actin: a novel mechanism regulating the subcellular localization of a major cytoskeletal protein. EMBO J. 17, 1635– 1741 (1998).
Medema, J. P.et al. FLICE is activated by association with the CD95 death-inducing signaling complex (DISC). EMBO J. 16, 2794 –2804 (1997).
Yang, X., Chang, H. Y. & Baltimore, D. Essential role of CED-4 oligomerization in CED-3 activation and apoptosis. Science 281, 1355– 1357 (1998).
Srinivasula, S. M., Ahmad, M., Fernandes-Alnemri, T. & Alnemri, E. S. Autoactivation of procaspase-9 by Apaf-1-mediated oligomerization. Mol. Cell 1, 949–957 ( 1998).
Liu, X., Zou, H., Slaughter, C. & Wang, X. DFF, a heterodimeric protein that functions downstream of caspase-3 to trigger DNA fragmentation during apoptosis. Cell 89, 175– 184 (1997).
Stegh, A. H.et al. DEDD, a novel death effector domain-containing protein, targeted to the nucleolus. EMBO J. 17, 5974– 5986 (1998).
Medema, J. P., Scaffidi, C., Krammer, P. H. & Peter, M. E. Bcl-xL acts downstream of caspase-8 activation by the CD95 death-inducing signaling complex. J. Biol. Chem. 273, 3388 –3393 (1998).
Li, H., Zhu, H., Xu, C. J. & Yuan, J. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94, 491–501 (1998).
Luo, X.et al. Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 94, 481–490 ( 1998).
Chinnaiyan, A. M., O'Rourke, K., Lane, B. R. & Dixit, V. M. Interaction of CED-4 with CED-3 and CED-9: a molecular framework for cell death. Science 275, 1122– 1126 (1997).
Hengartner, M. O. Apoptosis. CED-4 is a stranger no more. Nature 388, 714–715 (1997).
Yang, J.et al. Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 275, 1129– 1132 (1997).
Kluck, R. M., Bossy-Wetzel, E., Green, D. R. & Newmeyer, D. D. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275, 1132– 1136 (1997).
Pan, G., O'Rourke, K. & Dixit, V. M. Caspase-9, Bcl-XL, and Apaf-1 form a ternary complex. J. Biol. Chem. 273, 5841– 5845 (1998).
Hu, Y.et al. Bcl-XL interacts with Apaf-1 and inhibits Apaf-1-dependent caspase-9 activation. Proc. Natl Acad. Sci. USA 95, 4386– 4391 (1998).
Sakamaki, K., Tsukumo, S. & Yonehara, S. Molecular cloning and characterization of mouse caspase-8. Eur. J. Biochem. 253, 399– 405 (1998).
4. Sakamaki, K., Miyajima, I., Kitamura, T. & Miyajima, A. Critical cytoplasmic domains of the common beta subunit of the human GM- CSF, IL-3 and IL-5 receptors for growth signal transduction and tyrosine phosphorylation. EMBO J. 11, 3541–3549 (1992).
Nishimura, Y.et al. Expression and function of mouse Fas antigen on immature and mature T cells. J. Immunol. 154, 4395– 4403 (1995).
Eberstadt, M.et al. NMR structure and mutagenesis of the FADD (Mort1) death-effector domain. Nature 392, 941– 945 (1998).
van der Biezen, E. A. & Jones, J. D. The NB-ARC domain: a novel signaling motif shared by plant resistance gene products and regulators of cell death in animals. Curr. Biol. 8, R226–227 (1998).
Yonehara, S.et al. Involvement of apoptosis antigen Fas in clonal deletion of human thymocytes. Int. Immunol. 6, 1849– 1856 (1994).
Acknowledgements
We thank Y. Tsujimoto for cDNAs encoding Bcl-2 and SKW6.4 cells, D. V. Goeddel for mouse FADD cDNA, D. J. Pickup for CrmA cDNA, S. Hashimoto for antiserum against E1B19K, S.Tsukumo for pME18S-Flag2, H. Kazama for pME18S–Myc and FH2 cells, and Y. Nakanishi for HF1 cells. This work was supported in part by a Grant-in-Aid from the Ministry of Education, Science, Sports and Culture of Japan, and by the Ministry of Health and Welfare, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Imai, Y., Kimura, T., Murakami, A. et al. The CED-4-homologous protein FLASH is involved in Fas-mediated activation of caspase-8 during apoptosis. Nature 398, 777–785 (1999). https://doi.org/10.1038/19709
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/19709
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.