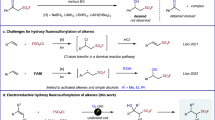
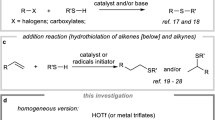
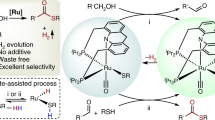
Abstract
THE identification of the keto-acid 2,4-dinitrophenylhydrazones is greatly facilitated by conversion to their respective amino-acids. The most commonly employed procedure is that first noted by Emil Fischer1 and modified for application to organic systems by Towers et al.2. This involves low-pressure hydrogenolysis over a platinum catalyst, followed by paper chromatography of the amino-acids produced. The yields of the respective amino-acids vary between 30 and 98 per cent, and in general the conversion is free from side-reactions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fischer, E., and Groh, R., Ann., 383, 363 (1911).
Towers, G. H. N., Thompson, J. F., and Steward, F. C., J. Amer. Chem. Soc., 76, 2392 (1954).
Menkes, J. H., A.M.A., J. Dis. Child., 99, 500 (1960).
Menkes, J. H., and Jervis, G. A., Pediatrics (in the press).
Smith, H. A., Alderman, D. M., and Nadig, F. W., J. Amer. Chem. Soc., 67, 272 (1945).
Waser, E., and Brauchli, E., Helv. Chim. Acta, 7, 740 (1924).
Levin, R. H., and Pendergrass, J. H., J. Amer. Chem. Soc., 69, 2436 (1947).
Smith, H. A., Catalysis, edit. by Emmett, P. H., 5, 175 (New York, 1957).
Edson, N. L., J. Soc. Chem. Indust., 53, 138 T (1934).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MENKES, J. Side-Reactions during Hydrogenolysis of 2,4-Dinitrophenylhydrazones of Aromatic Keto-Acids. Nature 191, 285–286 (1961). https://doi.org/10.1038/191285a0
Issue Date:
DOI: https://doi.org/10.1038/191285a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.