Abstract
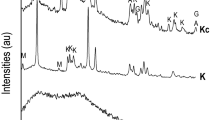
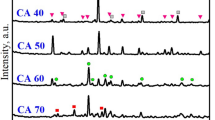
ALTHOUGH the physical effects of the carbonation of hydrated cements and mortars have been studied1–3, the way in which the carbon dioxide is held has not yet been clearly established. Most workers have assumed that it exists in the form of calcite ; but in work at this Division3,4 and elsewhere5 the quantity of calcite detected by X-ray diffraction and differential thermal methods is always much less than the amount of carbon dioxide recoverable from the samples. We have investigated this problem in some detail using samples of mortar and carbonated calcium silicate hydrate.
Similar content being viewed by others
Article PDF
References
Leber, I., and Blakey, F. A., J. Amer. Concr. Inst., 28, 295 (1956) (Proc. 53).
Verbeck, G. J., A.S.T.M. Spec. Tech. Pub. No. 205, 17 (1958).
Kroone, B., and Blakey, F. A. (unpublished results).
Cole, W. F. (unpublished results).
Gaze, R., and Robertson, R. H. S., Mag. Concr. Res., 8, 7 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
COLE, W., KROONE, B. Carbonate Minerals in Hydrated Portland Cement. Nature 184, BA57 (1959). https://doi.org/10.1038/184057a0b
Issue Date:
DOI: https://doi.org/10.1038/184057a0b
This article is cited by
-
A fully coupled Hydraulic Mechanical Chemical approach applied to cementitious material damage due to carbonation
npj Materials Degradation (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.