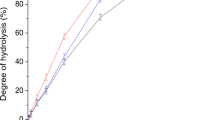
Abstract
THE synthesis of the chain-forming α-1: 4-bonds of potato starch (amylose + amylopectin) has for long been ascribed to phosphorylase acting on glucose-1-phosphate and a preformed starch-like primer1. We now report the synthesis of amylose-like material by another enzyme system from potato. This synthesis makes use of D-enzyme, discovered in 1953 by Peat and co-workers2. D-enzyme catalyses the reversible disproportionation of maltodextrins, the glucose oligosaccharides containing the α-1: 4-linkage. For example, maltotriose is acted on to give, as the first products of reaction, glucose and malto-pentaose, and at equilibrium glucose and a whole series of maltodextrins are present. However, with maltotriose as the initial substrate, none of the synthetic oligosaccharides is of sufficient length to form a coloured complex with iodine. The minimum length for colour formation is at least 12 glucose units3. Incubation of D-enzyme with a maltodextrin and glucose lowers the average length of the polymer because of transfer of some of its glucose residues to the added glucose. It was realized that a reversal of this procedure, the removal of glucose, should increase the length of the polymers and might ultimately result in the synthesis from a small maltodextrin molecule of an iodine-staining, amylose-like polysaccharide. If, in addition to D-enzyme, the potato is equipped with such a glucose-removing enzyme system, then it is presumably capable of synthesizing amylose by a route other than that provided by phosphorylase. A suitable glucose-removing system is hexokinase and adenosine tri-phosphate, and hexokinase is known to be in the potato4. This will convert glucose into glucose-6-phosphate, and D-enzyme does not transfer chain segments from maltodextrins to the sugar phosphate as it does to glucose5. The system (maltodextrin + D-enzyme + hexokinase + adenosine triphosphate) should therefore bring about amylose synthesis. This has been achieved.
Similar content being viewed by others
Article PDF
References
Hanes, C. S., Proc. Roy. Soc., B, 129, 174 (1940).
Peat, S., Whelan, W. J., and Rees, W. R., Nature, 172, 158 (1953); J. Chem. Soc., 44 (1956).
Bailey, J. M., and Whelan, W. J. (to be published).
Kotel'nikova, A. V., Biokhimiya, 17, 462 (1952). Saltman, P., J. Biol. Chem., 200, 145 (1953).
Peat, S., Whelan, W. J., and Jones, G., J. Chem. Soc., 2490 (1957).
Whelan, W. J., “Encyclopedia of Plant Physiology”, 6, 154 (Springer-Verlag, Berlin, 1958).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WALKER, G., WHELAN, W. Synthesis of Amylose by Potato D-Enzyme. Nature 183, 46 (1959). https://doi.org/10.1038/183046a0
Issue Date:
DOI: https://doi.org/10.1038/183046a0
This article is cited by
-
Pcal_0768, a hyperactive 4-α-glucanotransferase from Pyrobacculum calidifontis
Extremophiles (2016)
-
Carbohydrate‐active enzymes exemplify entropic principles in metabolism
Molecular Systems Biology (2011)
-
Recent Advances in the Biochemistry of Glycogen and Starch
Nature (1961)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.