Abstract
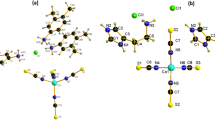
IN former communications1, we reported that the cupric salts, anhydrous or hydrated, with n- and iso-monocarboxylic acids might consist of the dimeric molecules having a sort of copper-to-copper linkage. In continuation of these researches, we have recently examined the structure of cupric salts with mono-, di- and tri-chloroacetic acids, using the criteria previously derived: (1) the cupric alkanoates involving the dimeric molecules show, in addition to a band at about 43 × 1013 c./s., an absorption band of a special kind at about 80 × 1013 c./s., which was regarded as due possibly to the presence of the copper-to-copper linkage; (2) the polarization of the latter band is the reverse of the polarization of the former.
Similar content being viewed by others
Article PDF
References
Tsuchida, R., and Yamada, S., Nature, 176, 1171 (1955). Tsuchida, R., Yamada, S., and Nakamura, H., ibid., 178, 1192 (1956); Bull. Chem. Soc. Japan (in the press).
Tsuchida, R., Kobayashi, M., Bull. Chem. Soc. Japan, 13, 619 (1938); “The Colour and the Structure of Metallic Compounds” (Osaka, 1944).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
TSUCHIDA, R., YAMADA, S. & NAKAMURA, H. Structure of Cupric Salts with Mono-, Di- and Tri-chloroacetic Acids. Nature 181, 479 (1958). https://doi.org/10.1038/181479b0
Issue Date:
DOI: https://doi.org/10.1038/181479b0
This article is cited by
-
EPR study of antiferromagnetic interactions inside pairs of cupric ions in copper monochloroacetate 2.5-hydrate
Il Nuovo Cimento B Series 10 (1966)
-
Nature of Cupric Alkanoate Solutions
Nature (1960)
-
Structure of Cupric Mono-, Di- and Tri-bromoacetates
Nature (1958)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.