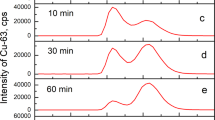
Abstract
INTEREST has been shown in the blue, copper-containing, serum oxidase cæruloplasmin, in particular because of its absence from the blood of subjects with hepatolenticular degeneration1. The copper of cæruloplasmin is firmly bound, being non-dialysable at neutral pH2. As part of a study of the mode of linkage of copper to the apo-enzyme, the effect of chymotryptic digestion on cæruloplasmin was investigated.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Scheinberg, I. H., and Gitlin, D., Science, 116, 484 (1952).
Holmberg, C. G., Laurell, C. B., Acta Chem. Scand., 2, 550 (1948).
Kekwick, R. A., and MacKay, M. E., Medical Research Council Special Report No. 286 (1954).
Kominz, D. R. (personal communication).
Mahler, H. R., Hubscher, G., and Baum, H., J. Biol. Chem., 216, 625 (1955).
Azari, P. R., and Feeney, R. E., Fed. Proc., 16, 147 (1957).
Scheinberg, I. H., and Morell, A. G., J. Clin. Invest., 36, 1193 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
CURZON, G. Effect of Chymotrypsin on Cæruloplasmin. Nature 181, 115–116 (1958). https://doi.org/10.1038/181115b0
Issue Date:
DOI: https://doi.org/10.1038/181115b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.