Abstract
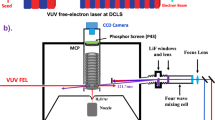
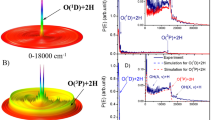
PREVIOUS work on the photolysis of ozone has led to the conclusion that a simple atom-chain mechanism is incapable of explaining the experimental facts. The existence of an energy chain involving excited oxygen molecules has gained wide acceptance due to the work of Schumacher1, Heidt and Forbes2, and Ritchie3. The nature of the excitation involved in the energy chain was not known, however, most workers assuming it to be the 1σ+g state of the oxygen4. Experiments on the flash photolysis of ozone5 have enabled us to show that the excitation is purely vibrational, the oxygen being formed in the ground electronic state with up to 16 quanta of vibrational energy, by reaction (1) which is exothermic to the extent of 93 kcal./mole for O(3P) and 138 kcal./mole for O(1D).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Schumacher, J. Amer. Chem. Soc., 52, 2377 (1930).
Heidt and Forbes, J. Amer. Chem. Soc., 56, 2365 (1934).
Ritchie, Proc. Roy. Soc., A, 146, 848 (1934).
Noyes and Leighton, “The Photochemistry of Gases” (Reinhold Publishing Corp., New York, 1941).
McGrath and Norrish, Proc. Roy. Soc., A, 242, 265 (1957).
Lipscomb, Norrish and Thrush, Proc. Roy. Soc., A, 233, 455 (1956).
McKinley, Garvin and Boudart, J. Chem. Phys., 23, 784 (1955).
Benson, J. Chem. Phys., 20, 1351 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McGRATH, W., NORRISH, R. Production of Vibrationally Excited Oxygen Molecules in the Flash Photolysis of Ozone. Nature 180, 1272–1273 (1957). https://doi.org/10.1038/1801272b0
Issue Date:
DOI: https://doi.org/10.1038/1801272b0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.