Abstract
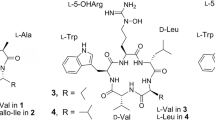
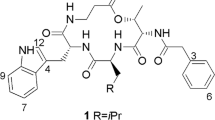
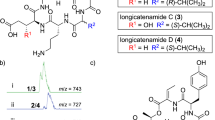
REPORTS in the literature1 regarding the dodecapeptide, bacitracin A, indicate the presence in the molecule of the sequences phenylalanyl histidine (I) and phenylalanyl isoleucyl cysteine (II). Two related facts remain unexplained: (a) there is only one phenylalanine residue in the molecule; hence it is the same phenylalanyl which is present in (I) and (II); (b) on total hydrolysis of dinitrophenyl–bacitracin A, the dinitrophenyl derivative of the isoleucine residue in (II) can be isolated, though in poor yield1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lockhart, I. M., and Abraham, E. P., Biochem. J., 58, 633 (1954). Lockhart, I. M., Abraham, E. P., and Newton, G. G. F., ibid., 61, 534 (1955). Hausmann, W., Weisiger, J. R., and Craig, L. C., J. Amer. Chem. Soc., 77, 723 (1955). Lockhart, I. M., and Abraham, E. P., Biochem. J., 62, 645 (1956).
Wrinch, D., Nature, 137, 411 (1936) et seq.; Proc. Roy. Soc., A, 160, 59 (1937) et seq.; Phil. Mag., 31, 183 (1941), etc.
Stoll, A., Hoffman, A., and Petrzilka, T., Helv. Chim. Acta, 34, 1544 (1951).
Stoll, A., “Prog. in Chem. of Org. Nat. Products”, 8, 114 (1952).
Cf. also Gaylord, N. G., Exper., 10, 351 (1954).
In this structure, R is C15H15N2, the lysergic acid residue. Residue two is L-phenylalanyl for ergotamine and ergocristine, L-leucyl for ergosine and ergokryptine and L-valyl for ergocornine. R1 is methyl for ergotamine and ergosine and CHMe2 in the three other cases. The third residue (prolyl in all cases) is also in the L-form.
Glenn, A. L., Quart. Rev. Chem. Soc., Lond., 8, 192 (1954).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WRINCH, D. Structure of Bacitracin A. Nature 179, 536–537 (1957). https://doi.org/10.1038/179536a0
Issue Date:
DOI: https://doi.org/10.1038/179536a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.