Abstract
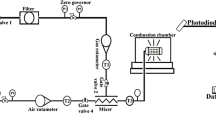
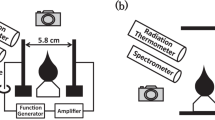
WHEN a stream of methyl nitrite vapour issuing from the mouth of an upright cylindrical glass tube about 5 cm. in diameter is ignited it burns in the air as a lambent diffusion flame, greyish-yellow in hue. At the inner surface of the diffusion flame an orange-red zone forms as a cone at the mouth of the tube. If the rate of flow is reduced, this orange-red inner flame peels away from the diffusion flame and slowly descends the tube as a flat disk surmounted by a fainter, conical afterglow of the same colour, easily visible in a darkened room. No oxygen is necessary for its propagation. After it has entered the tube it is not affected if the diffusion flame at the mouth is extinguished. It is the decomposition flame of pure methyl nitrite.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gray, P., Hall, A. R., and Wolfhard, H. G., Proc. Roy. Soc., A (in the press).
Gray, P., Proc. Roy. Soc., A, 221, 462 (1954).
Steacie, E. W. R., “Atom and Free Radical Reactions”, 2nd edit. (Reinhold, New York, 1954).
Rice, F. O., and Rodowskas, E. L., J. Amer. Chem. Soc., 57, 350 (1935).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
GRAY, P., HALL, A. & WOLFHARD, H. Decomposition Flame of Methyl Nitrite. Nature 176, 695–696 (1955). https://doi.org/10.1038/176695a0
Issue Date:
DOI: https://doi.org/10.1038/176695a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.