Abstract
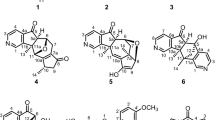
DURING a study of plant keto-acids carried out by one of us (G. H. N. T.) under the direction of Prof. F. C. Steward at Cornell University, an unknown substance, hereafter designated compound I, was found in the keto-acid fraction of all plant tissues examined1. This substance was isolated in crystalline form (sint. 206°, decomp. 214°) from the keto-acid dinitrophenylhydrazone fraction of Mentha piperita leaves by elution from an alumina column with 1 per cent sodium carbonate. Its position on chromatograms was similar to that of the slower-moving spot of pyruvic acid dinitrophenylhydrazone (Fig. 1, ref. 1). The pyruvic acid spot developed a chocolate colour when sprayed with alkali, whereas compound I gave a yellow colour. On hydrogenation, compound I yielded a ninhydrin-reactive substance (compound II) which was chromatographically similar to γ-amino-butyric acid (Fig. 2, ref. 1). The infra-red absorption spectrum of compound I did not display the carboxyl peak characteristic of α-keto-acid dinitrophenyl-hydrazoness.
Similar content being viewed by others
Article PDF
References
Towers, G. H. N., Thompson, J. F., and Steward, F. C., J. Amer. Chem. Soc., 76, 2392 (1954).
Brady, O. L., and Day, J. N. E., J. Chem. Soc., 123, 2266 (1923).
Isherwood, F. A., and Cruickshank, D. H., Nature, 173, 121 (1954).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
TOWERS, G., MORTIMER, D. Identification of an Artefact on Chromatograms of the Keto-acid 2,4-Dinitro-phenylhydrazones. Nature 174, 1189 (1954). https://doi.org/10.1038/1741189a0
Issue Date:
DOI: https://doi.org/10.1038/1741189a0
This article is cited by
-
Partition chromatography and its use in the plant sciences
The Botanical Review (1959)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.