Abstract
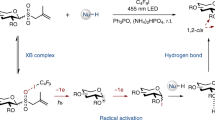
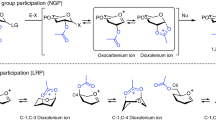
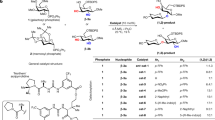
WHEN a sugar glycoside is oxidized with periodate, the ring is severed and a dialdehyde is formed1. In the case of pyranosides, cleavage is accompanied by the elimination of the —CH(OH)— group at C3 as formic acid, whereas in the cleavage of pentofuranosides no formic acid is formed. The elucidation of this reaction by recognition of the dialdehydic character of the periodate oxidation product1 and an extensive study of the corresponding dibasic acids and their salts have enabled the ring structure of glycosides to be determined and have provided a simple method for correlating the structure of the various glycosides2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fleury, Herissey and Joly, J. Pharm. Chim., 20, 149 (1934).
Hudson and Jackson, J. Amer. Chem. Soc., 58, 378 (1936); 59, 994 (1937).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
AKEER, M., CADOTTE, J., MONTGOMERY, R. et al. A New Procedure for Correlating the Structure of Glycosides. Nature 171, 474–475 (1953). https://doi.org/10.1038/171474a0
Issue Date:
DOI: https://doi.org/10.1038/171474a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.