Abstract
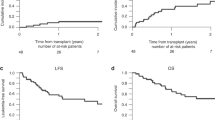
A regimen of busulfan and cyclophosphamide (BuCy2) is regarded as the standard myeloablative regimen for SCT. This study evaluated the hypothesis that fludarabine can replace cyclophosphamide for myeloablative allogeneic SCT. Ninety-five patients underwent allogeneic SCT from HLA-identical donors, following BuCy2 (n=55) or busulfan+fludarabine (BF, n=40). The efficacy of fludarabine compared to cyclophosphamide was retrospectively evaluated. The BF group exhibited a shorter duration until engraftment (P=0.001), lower incidence of acute and chronic GVHD (P<0.001 and P=0.003, respectively), and non-relapse mortality (NRM) (P=0.039). Furthermore, the event-free survival and overall survival were significantly higher for the BF group compared to the BuCy2 group (P=0.004 and 0.002, respectively). After adjusting for age, the risk status of disease, GVHD prophylaxis and donor type, the BF regimen was found to be an independent favorable risk factor for event-free survival (hazard ratio (HR), 0.181; 95% confidence interval, 0.045–0.720; P=0.016) and overall survival (HR, 0.168; 0.035–0.807; P=0.026). The replacement of cyclophosphamide with fludarabine for myeloablative conditioning seems to be more effective in terms of short-term NRM, and GVHD compared to BuCy2 regimen in allogeneic transplantation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
McDonald GB, Slattery JT, Bouvier ME, Ren S, Batchelder AL, Kalhorn TF et al. Cyclophosphamide metabolism, liver toxicity, and mortality following hematopoietic stem cell transplantation. Blood 2003; 101: 2043–2048.
Gandhi V, Plunkett W . Cellular and clinical pharmacology of fludarabine. Clin Pharmacokinet 2002; 41: 93–103.
Li L, Liu X, Glassman AB, Keating MJ, Stros M, Plunkett W et al. Fludarabine triphosphate inhibits nucleotide excision repair of cisplatin-induced DNA adducts in vitro. Cancer Res 1997; 57: 1487–1494.
Slavin S, Nagler A, Naparstek E, Kapelushnik Y, Aker M, Cividalli G et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood 1998; 91: 756–763.
Bornhauser M, Thiede C, Platzbecker U, Jenke A, Helwig A, Plettig R et al. Dose-reduced conditioning and allogeneic hematopoietic stem cell transplantation from unrelated donors in 42 patients. Clin Cancer Res 2001; 7: 2254–2262.
Murotani Y, Kuroda J, Kimura S, Terao K, Fukiya E, Ozawa M et al. Non-myeloablative haematopoietic stem cell transplantation for severe aplastic anaemia with various complications. Clin Lab Haematol 2002; 24: 303–306.
Or R, Shapira MY, Resnick I, Amar A, Ackerstein A, Samuel S et al. Nonmyeloablative allogeneic stem cell transplantation for the treatment of chronic myeloid leukemia in first chronic phase. Blood 2003; 101: 441–445.
de Lima M, Couriel D, Thall PF, Wang X, Madden T, Jones R et al. Once-daily intravenous busulfan and fludarabine: clinical and pharmacokinetic results of a myeloablative, reduced-toxicity conditioning regimen for allogeneic stem cell transplantation in AML and MDS. Blood 2004; 104: 857–864.
Bornhauser M, Storer B, Slattery JT, Appelbaum FR, Deeg HJ, Hansen J et al. Conditioning with fludarabine and targeted busulfan for transplantation of allogeneic hematopoietic stem cells. Blood 2003; 102: 820–826.
Russell JA, Tran HT, Quinlan D, Chaudhry A, Duggan P, Brown C et al. Once-daily intravenous busulfan given with fludarabine as conditioning for allogeneic stem cell transplantation: study of pharmacokinetics and early clinical outcomes. Biol Blood Marrow Transplant 2002; 8: 468–476.
Chunduri S, Dobogai LC, Peace D, Saunthararajah Y, Chen HY, Mahmud N et al. Comparable kinetics of myeloablation between fludarabine/full-dose busulfan and fludarabine/melphalan conditioning regimens in allogeneic peripheral blood stem cell transplantation. Bone Marrow Transplant 2006; 38: 477–482.
Boeckh M, Bowden RA, Goodrich JM, Pettinger M, Meyers JD . Cytomegalovirus antigen detection in peripheral blood leukocytes after allogeneic marrow transplantation. Blood 1992; 80: 1358–1364.
Sullivan KM, Shulman HM, Storb R, Weiden PL, Witherspoon RP, McDonald GB et al. Chronic graft-versus-host disease in 52 patients: adverse natural course and successful treatment with combination immunosuppression. Blood 1981; 57: 267–276.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant 1995; 15: 825–828.
Lee KH, Choi SJ, Lee JH, Lee JS, Kim WK, Lee KB et al. Prognostic factors identifiable at the time of onset of acute graft-versus-host disease after allogeneic hematopoietic cell transplantation. Haematologica 2005; 90: 939–948.
Sullivan KM, Agura E, Anasetti C, Appelbaum F, Badger C, Bearman S et al. Chronic graft-versus-host disease and other late complications of bone marrow transplantation. Semin Hematol 1991; 28: 250–259.
DeLeve LD . Hepatic venoocclusive disease: a major complication of hematopoietic stem cell transplantation in cancer patients. Tumori 2001; 87: S27–S29.
DeLeve LD . Cellular target of cyclophosphamide toxicity in the murine liver: role of glutathione and site of metabolic activation. Hepatology 1996; 24: 830–837.
Shulman HM, Luk K, Deeg HJ, Shuman WB, Storb R . Induction of hepatic veno-occlusive disease in dogs. Am J Pathol 1987; 126: 114–125.
Slattery JT, Kalhorn TF, McDonald GB, Lambert K, Buckner CD, Bensinger WI et al. Conditioning regimen-dependent disposition of cyclophosphamide and hydroxycyclophosphamide in human marrow transplantation patients. J Clin Oncol 1996; 14: 1484–1494.
Ferrara JL . Pathogenesis of acute graft-versus-host disease: cytokines and cellular effectors. J Hematother Stem Cell Res 2000; 9: 299–306.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chae, Y., Sohn, S., Kim, J. et al. New myeloablative conditioning regimen with fludarabine and busulfan for allogeneic stem cell transplantation: comparison with BuCy2. Bone Marrow Transplant 40, 541–547 (2007). https://doi.org/10.1038/sj.bmt.1705770
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705770
Keywords
This article is cited by
-
Fludarabine/busulfan versus busulfan/cyclophosphamide as myeloablative conditioning for myelodysplastic syndrome: a propensity score-matched analysis
Bone Marrow Transplantation (2021)
-
Pharmacokinetics of fludarabine and its association with clinical outcomes in paediatric haematopoietic stem cell transplantation patients
Bone Marrow Transplantation (2019)
-
Maximal concentration of intravenous busulfan as a determinant of veno-occlusive disease: a pharmacokinetic-pharmacodynamic analysis in 293 hematopoietic stem cell transplanted children
Bone Marrow Transplantation (2019)
-
Impact of in vivo T cell depletion in HLA-identical allogeneic stem cell transplantation for acute myeloid leukemia in first complete remission conditioned with a fludarabine iv-busulfan myeloablative regimen: a report from the EBMT Acute Leukemia Working Party
Journal of Hematology & Oncology (2017)
-
Busulfan fludarabine vs busulfan cyclophosphamide as a preparative regimen before allogeneic hematopoietic cell transplantation: systematic review and meta-analysis
Bone Marrow Transplantation (2016)