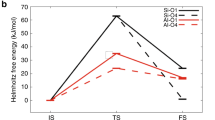
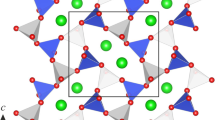
Abstract
SEVERAL polymorphic forms of anhydrous alumina derived from the calcination of hydrated aluminas have been described by various authors1–3. The interpretations made of the relationships which exist between the hydrated and anhydrous forms pre-suppose that the dehydration of gibbsite and bayerite occur through böhmite, and that the dehydration of the böhmite derived from gibbsite can unaccountably yield one of two series of transformations before α-alumina is finally produced.
Similar content being viewed by others
Article PDF
References
Weiser, H. B., and Milligan, W. O., J. Phys. Chem., 36, 3010 (1932); 38, 1175 (1934). Jellinek, M. H., and Fankuchen, I., Indust. and Eng. Chem., Indust. Edit., 37, 158 (1945).
Rooksby, H. P., “X-Ray Identifications and Crystal Structures of Clay Minerals”, 244 (Mineralogical Society, 1951).
Stumpf, H. C., Russell, A. S., et al., Indust. and Eng. Chem., 42, 7, 1398 (1950).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DAY, M., HILL, V. Thermal Transformations of the Aluminas and their Hydrates. Nature 170, 539 (1952). https://doi.org/10.1038/170539a0
Issue Date:
DOI: https://doi.org/10.1038/170539a0
This article is cited by
-
Dehydrating decomposition of alumina trihydrate
Die Naturwissenschaften (1958)
-
�ber Mechanismus und Verlauf der Entw�sserung von Hydrargillit und Bayerit
Die Naturwissenschaften (1957)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.