Summary:
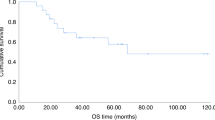
To evaluate the efficacy of reduced-intensity stem-cell transplantation (RIST), we retrospectively compared outcomes of 207 consecutive Japanese patients aged between 50 and 59 years with hematologic malignancies who received RIST (n=70) and conventional stem-cell transplantation (CST) (n=137). CST recipients received total body irradiation (TBI)-based or busulfan/cyclophosphamide-based regimens. RIST regimens were purine analog-based (n=67), 2 Gy TBI-based (n=2), and others (n=1). Most CST recipients (129/137) received calcineurin inhibitors and methotrexate as graft-versus-host (GVHD) prophylaxis, while 32 RIST recipients received cyclosporin. In all, 23 CST and five RIST recipients died without disease progression within 100 days of transplant. Grade II to IV acute GVHD occurred in 56 CST and 38 RIST recipients. There was no significant difference in overall survival (OS) and progression-free survival between CST and RIST. On multivariate analysis on OS, five variables were significant: preparative regimens (CST vs RIST) (hazard ratio=1.92, 95% confidence interval, 1.25–2.97; P=0.003), performance status (2–4 vs 0–1) (2.50, 1.51–4.16; P<0.001), risk of underlying diseases (1.85, 1.21–2.83; P=0.004), acute GVHD (2.57, 1.72–3.84; P<0.001), and CML (0.38, 0.21–0.69; P=0.002). We should be careful in interpreting results of this small-sized retrospective study; however, reduced regimen-related toxicity might contribute to better survival in RIST. The low relapse rates following RIST suggest a strong antitumor activity through allogeneic immunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Biggs JC, Horowitz MM, Gale RP et al. Bone marrow transplants may cure patients with acute leukemia never achieving remission with chemotherapy. Blood 1992; 80: 1090–1093.
Ratanatharathorn V, Karanes C, Lum LG et al. Allogeneic bone marrow transplantation in high-risk myeloid disorders using busulfan, cytosine arabinoside and cyclophosphamide (BAC). Bone Marrow Transplant 1992; 9: 49–55.
Bearman S, Appelbaum FR, Buckner C et al. Regimen-related toxicity in patients undergoing bone marrow transplantation. J Clin Oncol 1988; 6: 1562–1568.
Slavin S, Nagler A, Naparstek E et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood 1998; 91: 756–763.
Giralt S, Estey E, Albitar M et al. Engraftment of allogeneic hematopoietic progenitor cells with purine analog-containing chemotherapy: harnessing graft-versus-leukemia without myeloablative therapy. Blood 1997; 89: 4531–4536.
Saito T, Kanda Y, Kami M et al. Therapeutic potential of a reduced-intensity preparative regimen for allogeneic transplantation with cladribine, busulfan, and antithymocyte globulin against advanced/refractory acute leukemia/lymphoma. Clin Cancer Res 2002; 8: 1014–1020.
Michallet M, Bilger K, Garban F et al. Allogeneic hematopoietic stem-cell transplantation after nonmyeloablative preparative regimens: impact of pretransplantation and posttransplantation factors on outcome. J Clin Oncol 2001; 19: 3340–3349.
Nagler A, Slavin S, Varadi G et al. Allogeneic peripheral blood stem cell transplantation using a fludarabine-based low intensity conditioning regimen for malignant lymphoma. Bone Marrow Transplant 2000; 25: 1021–1028.
Diaconescu R, Flowers CR, Storer B et al. Morbidity and mortality with nonmyeloablative compared with myeloablative conditioning before hematopoietic cell transplantation from HLA-matched related donors. Blood 2004; 104: 1550–1558.
Imataki O, Kami M, Kim SW et al. A nationwide survey of deep fungal infections and fungal prophylaxis after hematopoietic stem cell transplantation in Japan. Bone Marrow Transplant 2004; 33: 1173–1179.
Bacigalupo A . Second EBMT Workshop on reduced intensity allogeneic hemopoietic stem cell transplants (RI-HSCT). Bone Marrow Transplant 2002; 29: 191–195.
Bacigalupo A . Third EBMT/AMGEN Workshop on reduced-intensity conditioning allogeneic haemopoietic stem cell transplants (RIC-HSCT), and panel consensus. Bone Marrow Transplant 2004; 33: 691–696.
Gooley TA, Leisenring W, Crowley J, Storer BE . Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 1999; 18: 695–706.
Yamamoto R, Kusumi E, Kami M et al. Late hemorrhagic cystitis after reduced-intensity hematopoietic stem cell transplantation (RIST). Bone Marrow Transplant 2003; 32: 1089–1095.
Hogan WJ, Maris M, Storer B et al. Hepatic injury after nonmyeloablative conditioning followed by allogeneic hematopoietic cell transplantation: a study of 193 patients. Blood 2004; 103: 78–84.
Fukuda T, Hackman RC, Guthrie KA et al. Risks and outcomes of idiopathic pneumonia syndrome after nonmyeloablative and conventional conditioning regimens for allogeneic hematopoietic stem cell transplantation. Blood 2003; 102: 2777–2785.
Fukuda T, Boeckh M, Carter RA et al. Risks and outcomes of invasive fungal infections in recipients of allogeneic hematopoietic stem cell transplants after nonmyeloablative conditioning. Blood 2003; 102: 827–833.
McSweeney PA, Niederwieser D, Shizuru JA et al. Hematopoietic cell transplantation in older patients with hematologic malignancies: replacing high-dose cytotoxic therapy with graft-versus-tumor effects. Blood 2001; 97: 3390–3400.
Nakai K, Mineishi S, Kami M et al. Antithymocyte globulin affects the occurrence of acute and chronic graft-versus-host disease after a reduced-intensity conditioning regimen by modulating mixed chimerism induction and immune reconstitution. Transplantation 2003; 75: 2135–2143.
Mielcarek M, Martin PJ, Leisenring W et al. Graft-versus-host disease after nonmyeloablative versus conventional hematopoietic stem cell transplantation. Blood 2003; 102: 756–762.
Fontaine P, Roy-Proulx G, Knafo L et al. Adoptive transfer of minor histocompatibility antigen-specific T lymphocytes eradicates leukemia cells without causing graft-versus-host disease. Nat Med 2001; 7: 789–794.
Socie G, Stone JV, Wingard JR et al. Long-term survival and late deaths after allogeneic bone marrow transplantation. Late Effects Working Committee of the International Bone Marrow Transplant Registry. N Engl J Med 1999; 341: 14–21.
Gomez-Nunez M, Martino R, Caballero MD et al. Elderly age and prior autologous transplantation have a deleterious effect on survival following allogeneic peripheral blood stem cell transplantation with reduced-intensity conditioning: results from the Spanish multicenter prospective trial. Bone Marrow Transplant 2004; 33: 477–482.
Tallman MS, Kopecky KJ, Amos D et al. Analysis of prognostic factors for the outcome of marrow transplantation or further chemotherapy for patients with acute nonlymphocytic leukemia in first remission. J Clin Oncol 1989; 7: 326–337.
Hansen JA, Gooley TA, Martin PJ et al. Bone marrow transplants from unrelated donors for patients with chronic myeloid leukemia. N Engl J Med 1998; 338: 962–968.
Bolwell BJ . Are predictive factors clinically useful in bone marrow transplantation? Bone Marrow Transplant 2003; 32: 853–861.
Acknowledgements
This study was supported by a Grant-in-aid of the Ministry of Labor and Welfare in Japan. We thank T Fukuda for scientific discussions and for critically reviewing the manuscript. We thank all the staff and resident members of the transplant centers in Japan. A complete list of participating institutions appears in the Appendix.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
This study was conducted at the following institutions under the auspices of the following investigators in Japan: Tanimoto E Tetsuya (Kyusyu University Graduate School of Medical Sciences, Fukuoka), Iida H (Meitetsu Hospital, Aichi), Matsue K (Kameda General Hospital, Chiba), Kato K (Hamanomachi Hospital, Fukuoka), Shinagawa K (Okayama University Medical School, Okayama), Abe Y (Kyusyu University Graduate School of Medical Sciences, Fukuoka), Nakajyo T (Kanazawa University Graduate School of Medicine, Kanazawa), Uike N (National Kyushu Cancer Center, Fukuoka), Okamoto S (Keio University School of Medicine, Tokyo), Hirabayashi N (Nagoya Daini Red Cross Hospital, Aichi), Komatsu T (Tsukuba Memorial Hospital, Ibaraki), Tamaki S (Yamada Red Cross Hospital, Mie), Izumi Y (Kokura Memorial Hospital, Fukuoka), Karasuno T (Osaka Medical Center for Cancer and Cardiovascular Diseases, Osaka), Yamane T (Osaka City University, Osaka), Ashida T (Kinki University Hospital, Osaka), Wakita A (Nagoya City University Graduate School of Medical Science, Aichi), Furukawa T (Niigata Uniersity Medical Hospital, Niigata), Teshima H (Osaka City General Hospital, Osaka), Yamashita T (National Defense Medical College Hospital, Saitama), Miyazaki Y (Kansai Medical University Hospital, Osaka), Kobayashi Y & Taniwaki M (Kyoto Prefectural University of Medicine, Kyoto), Kobayashi H (Nagano Red Cross Hospital, Nagano), Ito T (Nihon University School of Medicine, Tokyo), Ishida Y (Iwate Medical University Hospital, Iwate), Yoshihara S (Osaka University Graduate School of Medicine, Osaka), Ri M (Shizuoka Saiseikai General Hospital, Shizuoka), Fukushima N (Saga Medical School, Saga), Iwashige A (University of Occupational and Environmental Health, Fukuoka), Togitani K (Kochi Medical School, Kochi), Yamamoto Y (Kishiwada City Hospital, Osaka), Otsuka E (Oita Medical University, Oita), Fujiyama Y (Shiga University of Medical Science, Shiga), Hirokawa M (Akita University School of Medicine, Akita), Nishimura M (Chiba University Graduate School of Medicine, Chiba), Imamura S (Fukui Medical University, Fukui), Masauzi N (Hakodate Municipal Hospital, Hokkaido), Hara M (Ehime Prefectural Central Hospital, Ehime), Moriuchi Y (Sasebo City General Hospital, Nagasaki), Hamaguchi M (Nagoya National Hospital, Aichi), Nishiwaki K (The JikeiUniversity School of Medicine, Tokyo), Yokota A (Chiba Municipal Hospital, Chiba), Takamatsu Y (Fukuoka University School of Medicine, Fukuoka).
Rights and permissions
About this article
Cite this article
Kojima, R., Kami, M., Kanda, Y. et al. Comparison between reduced intensity and conventional myeloablative allogeneic stem-cell transplantation in patients with hematologic malignancies aged between 50 and 59 years. Bone Marrow Transplant 36, 667–674 (2005). https://doi.org/10.1038/sj.bmt.1705122
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705122
Keywords
This article is cited by
-
Cardiovascular events among recipients of hematopoietic stem cell transplantation-A systematic review and meta-analysis
Bone Marrow Transplantation (2023)
-
Indications and outcomes of reduced-toxicity hematopoietic stem cell transplantation in adult patients with hematological malignancies
International Journal of Hematology (2013)
-
Role of allo-SCT for CML in 2010
Bone Marrow Transplantation (2010)
-
Favorable outcomes of intravenous busulfan, fludarabine, and 400 cGy total body irradiation-based reduced-intensity conditioning allogeneic stem cell transplantation for acute myelogenous leukemia with old age and/or co-morbidities
International Journal of Hematology (2010)
-
The evolution of hematopoietic SCT in myelodysplastic syndrome
Bone Marrow Transplantation (2009)