Summary:
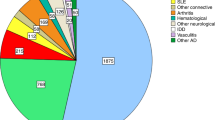
Experimental data and early phase I/II studies suggest that high-dose chemotherapy followed by autologous hematopoietic stem cell transplantation (HSCT) can arrest progression of severe autoimmune diseases. We have evaluated the toxicity and disease response in 473 patients with severe autoimmune disease treated with autologous HSCT between 1995 and 2003, from 110 centers participating in the European Group for Blood and Marrow Transplantation (EBMT) autoimmune disease working party database. Survival, transplant-related mortality, treatment response and disease progression were assessed. In all, 420 patients (89%; 86±4% at 3 years, median follow-up 20 months) were alive, 53 (11%) had died from transplant-related mortality (N=31; 7±3% at 3 years) or disease progression (N=22; 9±4% at 3 years). Of 370 patients, 299 evaluable for response (81%) showed a treatment response, which was sustained in 213 (71% of responders). Response was associated with disease (P<0.001), was better in patients who received cyclophosphamide during mobilization (relative risk (RR)3.28 (1.57–6.83)) and was worse with increasing age (>40 years, RR0.29 (0.11–0.82)). Disease progression was associated with disease (P<0.001) and conditioning intensity (high intensity, RR1; intermediate intensity, RR1.81 (0.96–3.42)); low intensity, RR2.34 (1.074–5.11)). These data from the collective EBMT experience support the hypothesis that autologous HSCT can alter disease progression in severe autoimmune disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Davidson A, Diamond B . Autoimmune diseases. N Engl J Med 2001; 345: 340–350.
Gratwohl A, Passweg J, Gerber I, Tyndall A . Stem cell transplantation for autoimmune diseases. Best Pract Res Clin Haematol 2001; 14: 755–776.
Marmont AM . Stem cell transplantation for autoimmune disorders. Coincidental autoimmune disease in patients transplanted for conventional indications. Best Pract Res Clin Haematol 2004; 17: 223–232.
van Bekkum DW . Stem cell transplantation for autoimmune disorders. Preclinical experiments. Best Pract Res Clin Haematol 2004; 17: 201–222.
Ikehara S . Treatment of autoimmune diseases by hematopoietic stem cell transplantation. Exp Hematol 2001; 29: 661–669.
Burt RK, Slavin S, Burns WH, Marmont AM . Induction of tolerance in autoimmune diseases by hematopoietic stem cell transplantation: getting closer to a cure? Int J Hematol 2002; 76 (Suppl. 1): 226–247.
Marmont A, Tyndall A, Gratwohl A, Vischer T . Haemopoietic precursor-cell transplants for autoimmune diseases. Lancet 1995; 345: 978.
Tyndall A, Gratwohl A . Haemopoietic stem and progenitor cells in the treatment of severe autoimmune diseases. Ann Rheum Dis 1996; 55: 149–151.
Van Laar JM, Tyndall A . Intense immunosuppression and stem-cell transplantation for patients with severe rheumatic autoimmune disease: a review. Cancer Control 2003; 10: 57–65.
Binks M, Passweg JR, Furst D et al. Phase I/II trial of autologous stem cell transplantation in systemic sclerosis: procedure related mortality and impact on skin disease. Ann Rheum Dis 2001; 60: 577–584.
Farge D, Marolleau JP, Zohar S et al. Autologous bone marrow transplantation in the treatment of refractory systemic sclerosis: early results from a French multicentre phase I–II study. Br J Haematol 2002; 119: 726–739.
Fassas A, Passweg JR, Anagnostopoulos A et al. Hematopoietic stem cell transplantation for multiple sclerosis. A retrospective multicenter study. J Neurol 2002; 249: 1088–1097.
Wulffraat NM, Sanders LA, Kuis W . Autologous hemopoietic stem-cell transplantation for children with refractory autoimmune disease. Curr Rheumatol Rep 2000; 2: 316–323.
Traynor AE, Barr WG, Rosa RM et al. Hematopoietic stem cell transplantation for severe and refractory lupus. Analysis after five years and fifteen patients. Arthritis Rheum 2002; 46: 2917–2923.
Rosen O, Thiel A, Massenkeil G et al. Autologous stem-cell transplantation in refractory autoimmune diseases after in vivo immunoablation and ex vivo depletion of mononuclear cells. Arthritis Res 2000; 2: 327–336.
Kashyap A, Passweg J, Tyndall A . Autologous hematopoietic stem cell transplant conditioning regimens for the treatment of severe autoimmune diseases. In: Dicke KA, Keating A (eds). Autologous Blood and Marrow Transplantation. Proceedings of the Ninth international Symposium. Carden Jennings Publishing: Arlington, Texas, Charlottesville, 2001.
Snowden JA, Passweg J, Moore JJ et al. Autologous hemopoietic stem cell transplantation in severe rheumatoid arthritis: a report from the EBMT and ABMTR. J Rheumatol 2004; 31: 482–488.
Wulffraat NM, Brinkman D, Ferster A et al. Long-term follow-up of autologous stem cell transplantation for refractory juvenile idiopathic arthritis. Bone Marrow Transplant 2003; 32 (Suppl. 1): S61–S64.
Jayne D, Passweg J, Marmont A et al. Autologous stem cell transplantation for systemic lupus erythematosus. Lupus 2004; 13: 168–176.
Huhn RD, Fogarty PF, Nakamura R et al. High-dose cyclophosphamide with autologous lymphocyte-depleted peripheral blood stem cell (PBSC) support for treatment of refractory chronic autoimmune thrombocytopenia. Blood 2003; 101: 71–77.
Durez P, Toungouz M, Schandene L et al. Remission and immune reconstitution after T-cell-depleted stem-cell transplantation for rheumatoid arthritis. Lancet 1998; 352: 881.
McColl G, Kohsaka H, Szer J, Wicks I . High-dose chemotherapy and syngeneic hemopoietic stem-cell transplantation for severe, seronegative rheumatoid arthritis. Ann Intern Med 1999; 131: 507–509.
Verburg RJ, Kruize AA, van den Hoogen FH et al. High-dose chemotherapy and autologous hematopoietic stem cell transplantation in patients with rheumatoid arthritis: results of an open study to assess feasibility, safety, and efficacy. Arthritis Rheum 2001; 44: 754–760.
Kim KC, Lee IH, Choi JH et al. Autologous stem cell transplantation in the treatment of refractory rheumatoid arthritis. J Korean Med Sci 2002; 17: 129–132.
Moore J, Brooks P, Milliken S et al. A pilot randomized trial comparing CD34-selected versus unmanipulated hemopoietic stem cell transplantation for severe, refractory rheumatoid arthritis. Arthritis Rheum 2002; 46: 2301–2309.
Lanza F, Dominici M, Govoni M et al. Prolonged remission state of refractory adult onset Still's disease following CD34-selected autologous peripheral blood stem cell transplantation. Bone Marrow Transplant 2000; 25: 1307–1310.
Wulffraat N, van Royen A, Bierings M et al. Autologous haemopoietic stem-cell transplantation in four patients with refractory juvenile chronic arthritis. Lancet 1999; 353: 550–553.
Quartier P, Prieur AM, Fischer A . Haemopoietic stem-cell transplantation for juvenile chronic arthritis. Lancet 1999; 353: 1885–1886.
Tyndall A, Black C, Finke J et al. Treatment of systemic sclerosis with autologous haemopoietic stem cell transplantation. Lancet 1997; 349: 254.
Shaughnessy PJ, Ririe DW, Ornstein DL et al. Graft failure in a patient with systemic lupus erythematosus (SLE) treated with high-dose immunosuppression and autologous stem cell rescue. Bone Marrow Transplant 2001; 27: 221–224.
Wulffraat NM, Sanders EA, Kamphuis SS et al. Prolonged remission without treatment after autologous stem cell transplantation for refractory childhood systemic lupus erythematosus. Arthritis Rheum 2001; 44: 728–731.
Myllykangas-Luosujarvi R, Jantunen E, Kaipiainen-Seppanen O et al. Autologous peripheral blood stem cell transplantation in a patient with severe mixed connective tissue disease. Scand J Rheumatol 2000; 29: 326–327.
Baron F, Ribbens C, Kaye O et al. Effective treatment of Jo-1-associated polymyositis with T-cell-depleted autologous peripheral blood stem cell transplantation. Br J Haematol 2000; 110: 339–342.
Bingham S, Griffiths B, McGonagle D et al. Autologous stem cell transplantation for rapidly progressive Jo-1-positive polymyositis with long-term follow-up. Br J Haematol 2001; 113: 840–841.
Fassas A, Anagnostopoulos A, Kazis A et al. Autologous stem cell transplantation in progressive multiple sclerosis – an interim analysis of efficacy. J Clin Immunol 2000; 20: 24–30.
Carreras E, Saiz A, Marin P et al. CD34+ selected autologous peripheral blood stem cell transplantation for multiple sclerosis: report of toxicity and treatment results at one year of follow-up in 15 patients. Haematologica 2003; 88: 306–314.
Openshaw H, Lund BT, Kashyap A et al. Peripheral blood stem cell transplantation in multiple sclerosis with busulfan and cyclophosphamide conditioning: report of toxicity and immunological monitoring. Biol Blood Marrow Transplant 2000; 6: 563–575.
Kozak T, Havrdova E, Pit'ha J et al. High-dose immunosuppressive therapy with PBPC support in the treatment of poor risk multiple sclerosis. Bone Marrow Transplant 2000; 25: 525–531.
Attal M, Harousseau JL, Facon T et al. Single versus double autologous stem-cell transplantation for multiple myeloma. N Engl J Med 2003; 349: 2495–2502.
Milpied N, Deconinck E, Gaillard F et al. Initial treatment of aggressive lymphoma with high-dose chemotherapy and autologous stem-cell support. N Engl J Med 2004; 350: 1287–1295.
Philip T, Guglielmi C, Hagenbeek A et al. Autologous bone marrow transplantation as compared with salvage chemotherapy in relapses of chemotherapy-sensitive non-Hodgkin's lymphoma. N Engl J Med 1995; 333: 1540–1545.
van Laar JM, McSweeney PA . High-dose immunosuppressive therapy and autologous progenitor cell transplantation for systemic sclerosis. Best Pract Res Clin Haematol 2004; 17: 233–245.
Mancardi GL, Saccardi R, Filippi M et al. Autologous hematopoietic stem cell transplantation suppresses Gd-enhanced MRI activity in MS. Neurology 2001; 57: 62–68.
Schmitz N, Linch DC, Dreger P et al. Randomised trial of filgrastim-mobilised peripheral blood progenitor cell transplantation versus autologous bone-marrow transplantation in lymphoma patients. Lancet 1996; 347: 353–357.
Openshaw H, Stuve O, Antel JP et al. Multiple sclerosis flares associated with recombinant granulocyte colony-stimulating factor. Neurology 2000; 54: 2147–2150.
Farge D, Passweg J, van Laar JM et al. Autologous stem cell transplantation in the treatment of systemic sclerosis: report from the EBMT/EULAR Registry. Ann Rheum Dis 2004; 63: 974–981.
Passweg JR, Rabusin M, Musso M et al. Haematopoietic stem cell transplantation for refractory autoimmune cytopenia. Br J Haematol 2004; 125: 749–755.
Petri M, Jones RJ, Brodsky RA . High-dose cyclophosphamide without stem cell transplantation in systemic lupus erythematosus. Arthritis Rheum 2003; 48: 166–173.
Storb RF, Lucarelli G, McSweeney PA, Childs RW . Hematopoietic cell transplantation for benign hematological disorders and solid tumors. Hematology (Am Soc Hematol Educ Program) 2003; 1: 372–397.
Tisdale JF, Dunn DE, Geller N et al. High-dose cyclophosphamide in severe aplastic anaemia: a randomised trial. Lancet 2000; 356: 1554–1559.
Acknowledgements
We would like to acknowledge the participating centres, the support of the project by the EBMT corporate members (Amgen Europe, Hoffmann-La Roche Ltd, Gilead Sciences, Baxter Oncology, Pharmacia Corporation, Chugai-Aventis, Fresenius HemoCare, SangStat, Schering AG, Gambro BCT, Elan Pharmaceuticals, Miltenyl Biotec GmbH, Therakos, Wyeth-Lederlé, Astra, Cobe International, Nextar, Liposome Co., Imtix, the Horton Foundation, the Swiss National Research Foundation, the Regional Cancer League Basel and the Délégation Régionale à la Recherche Clinique (DRRC), Assistance Publique-Hôpitaux de Paris (AP-HP), the French Ministry of Health (Programme Hospitalier de Recherche Clinique: PHRC 1997 AOM 97-030) and Professor E Gluckman, Hôpital St Louis, Paris).
Author information
Authors and Affiliations
Consortia
Corresponding author
Appendix A1
Appendix A1
List of participating centers:
Austria: Vienna, Vienna University Hospital; Australia: Brisbane, Mater Hospital; Hobart, Royal Hobart Hospital; Melbourne, Royal Melbourne Hospital; Perth, Sir Charles Gairdner Hospital; Royal Perth Hospital; Sydney, St Vincent's Hospital; Belgium: Brussels, Children University Hospital, Erasmus Hospital, Institut Jules Bordet; Liège, Centre Hospitalier Universitaire; Leuven, University Hospital Gasthuisberg; China: Nanjing, Gu Lou Hospital; Peking, Peking University Medical Center Hospital; Zhengzhou, The Third People Hospital of Zhengzhou; Czech Republic: Pilsen, Charles University Hospital; Prague, University Hospital Kralovske Vinohrady, University Hospital Motol; Finland: Kuopio, Kuopio University Hospital; Turku, Turku University Central Hospital; France: Besançon, Hôpital Jean Minijoz; Brest, Hôpital Morvan; Clermont Ferrant, Centre Hospitalier Universitaire; Lille, Hôpital Claude Huriez; Limoges, Centre Hospitalier Regional Universitaire Limoges; Marseilles, Institut Paoli Calmettes; Paris, Centre Hospitalier Universitaire Cochin, Hôpital Necker, Hôpital St Antoine, Hôpital St Louis, Pitie-Salpetriere; Strassbourg, Hôpital Universitaire de Hautepierre; Toulouse, Hôpital de Purpan; Germany: Berlin, Universitätsklinikum Charité, Charité-Virchow Klinikum; Bochum, University Hospital Knappschfts-Krankenhaus; Dresden, Universitätsklinikum; Düsseldorf, Henrich Heine Universität; Erlangen, University Hospital; Frankfurt, Universitätsklinikum; Freiburg, Klinikum der Albert-Ludwigs Universität Freiburg; Halle, Martin-Luther-Universität; Hannover, Medizinische Hochschule; Heidelberg, University of Heidelberg; Jena, University of Jena; Münster, Universitätsklinik; Tübingen, Medizinische Universitätsklinik; Wiesbaden, Deutsche Zentrum für Krankeitsdiagnostik; Hungary: Miskolc, Postgraduate Medical School; Greece: Thessaloniki, George Papanicolaou Hospital; Israel: Haifa, Rambam Medical Center; Jerusalem, Hadassah University Hospital; Italy: Cagliari, University of Cagliari; Ferrara, Ospedale St Anna; Firenze, Ospedale di Careggi, Ospedale Pediatrico ‘A.Meyer’; Genova, Ospedale San Martino; Milano, Ospedale San Raffaele; Modena, University of Modena; Monza, Ospedale San Gerardo; Torino, Ospedale San Luigi Orbassano; Padova, University Hospital; Palermo, Ospedale Oncologico ‘La Maddalena’; Pavia, Policlinico San Matteo; Pescara, Ospedale Civile; Pisa, University of Pisa; Reggio Emilia, Arcispedale Santa Maria Nuova; Reggio Calabria, Azienda Ospedaliera Bianchi-Melacrino-Morelli; Roma, Ospedale San Camillo, Università ‘La Sapienza’, Università Tor Vergata; Trieste, Istituto per l’Infanzia Burlo Garofolo; Udine, University Hospital; Japan: Osaka, Osaka Medical Center; The Netherlands: Leiden, Leiden University Medical Center; Nijmegen, University Medical Center St Radboud; Rotterdam, Erasmus University Medical Center; Utrecht, University Medical Center; Poland: Poznan, K. Marcinkowski University; Russia: Novosibirsk, Institute of Clinical Immunology; St Petersburg, St Petersburg State I. Pavlov Medical University; Slovakia: Bratislava, University Hospital; Spain: Barcelona, Hospital Clinic, Hospital Santa Creu i Sant Pau, Hospital Vall d'Hebron; Cádiz , Hospital del SAS; Lugo, Hospital Xeral-Calde; Madrid, Clinica Puerta de Hierro, Hospital Infantil La Paz, Hospital G.U. Gregorio Maranon, Hospital de la Princesa; Malaga, Hospital Carlos Haya; Palma de Mallorca, Hospital San Dureta; Santander, Hospital Universitario ‘Marquéz de Valdecilla’; Sevilla, Hospital Universitario Virgen del Rocio; Valencia, Hospital Clinico Universitario; Sweden: Göteborg, Sahlgrenska University Hospital; Lund, University Hospital; Uppsala, University Hospital; Switzerland: Basel, Kantonsspital; Zürich, University Children Hospital; UK: Birmingham, University of Birmingham; Leeds, University of Leeds; Liverpool, Royal Liverpool University Hospital; London, Great Ormond Street Hospital, Hammersmith Hospital, Royal Free Hospital, St George's Hospital, University College Hospital; Newcastle, Royal Victoria Infirmary; Nottingham, Nottingham City Hospital.
Rights and permissions
About this article
Cite this article
Gratwohl, A., Passweg, J., Bocelli-Tyndall, C. et al. Autologous hematopoietic stem cell transplantation for autoimmune diseases. Bone Marrow Transplant 35, 869–879 (2005). https://doi.org/10.1038/sj.bmt.1704892
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704892
Keywords
This article is cited by
-
Stem Cell Therapy as a Treatment for Autoimmune Disease—Updates in Lupus, Scleroderma, and Multiple Sclerosis
Current Allergy and Asthma Reports (2021)
-
Protective effects of mesenchymal stem cells on ischemic brain injury: therapeutic perspectives of regenerative medicine
Cell and Tissue Banking (2021)
-
Hematopoietic Stem Cell Transplantation for Systemic Sclerosis: Review of Current Status
BioDrugs (2019)
-
Autologous haematopoietic stem cell transplantation (AHSCT) in autoimmune disease adult patients in France: analysis of the long-term outcome from the French Society for Bone Marrow Transplantation and Cellular Therapy (SFGM-TC)
Clinical Rheumatology (2019)
-
Safety analysis in patients with autoimmune disease receiving allogeneic mesenchymal stem cells infusion: a long-term retrospective study
Stem Cell Research & Therapy (2018)